Biological cells undergo a process known as ’heat stress response’ when exposed to excessively high temperatures. Heat stress triggers a cascade of signaling mechanisms to produce a variety of heat shock proteins (Hsps), including small heat shock proteins (sHsps).
These ’cellular firefighters’ protect heat-sensitive structures, prevent proteins from clumping, and help refold damaged proteins.
In Escherichia coli, an sHsp called IbpA serves an interesting additional purpose. Besides its conventional role as a chaperone, IbpA also acts as a ’safety catch’ that suppresses its own synthesis when it is unnecessary.
Simply put, IbpA can regulate the expression of specific genes in response to temperature cues by affecting their translation from messenger RNA to protein. Although scientists recently found that IbpA regulates its own expression and that of its paralog (a gene that shares a common ancestor and arises from gene duplication events) IbpB, it is still unclear whether this protein regulates other genes as well.
Knowledge gap
In a recent study published in PNAS, Prof. Hideki Taguchi and Dr. Tsukumi Miwa from Tokyo Institute of Technology (Tokyo Tech) in Japan have addressed this knowledge gap. The duo explored, through various experiments with genetically engineered E. coli strains, the mechanisms via which IbpA regulates the heat shock response, revealing a new pivotal function of the Hsp.
They initially found that overexpression of IbpA led to a significant downregulation of many Hsps. Further experiments showed that IbpA has no effect on these Hsps at the translational level, i.e., it does not stop the synthesis of these proteins from RNA.
This led the researchers to hypothesize that IbpA may be influencing the expression of σ32, a master heat shock transcription factor responsible for regulating the expression of around a hundred genes, many of which are related to the heat shock response.
New model
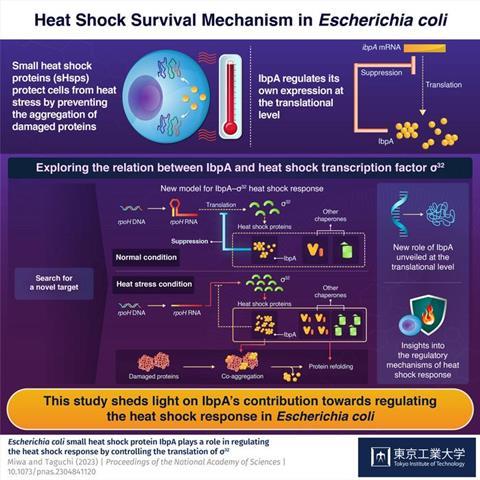
Through experiments involving IbpA-overexpressing and IbpA-deleted mutants, the researchers found that IbpA suppresses the expression of σ32 at the translational rather than transcriptional level. Accordingly, they proposed a new model explaining how IbpA operates under normal and heat stress conditions.
In this model, when the temperature is not excessively high, IbpA suppresses the translation of rpoH RNA that produces σ32. In turn, the lack of σ32 keeps the levels of Hsps, including IbpA, low.
However, when heat shock occurs, IbpA binds to misfolded or clumped-up proteins, leaving rpoH RNA alone. This leads to a rapid increase in the amount of σ32, which activates the production of Hsps to deal with the damage caused by heat stress.
Physiological significance
These results, however, beg the question: what is the physiological significance of IbpA suppressing rpoH translation? “One possibility is that the regulation of expression at the translation level is generally rapid and thus well-suited for immediate induction upon heat shock,” explains Prof. Taguchi.
“Another advantage of tight suppression of the σ32 levels by IbpA under normal conditions is the suppression of Hsp expression, which is unnecessary in the absence of stress.”
Overall, this study helps clarify how bacterial species adapt to harsh environments.
Prof. Taguchi remarks: “Our study focusing on IbpA has shed new light on the involvement of this previously unrecognized factor in the heat shock response of E. coli.” This knowledge could prove essential in bioengineering, industrial, medical, and environmental applications.





No comments yet