Photosynthetic bacteria, unlike plants, do not generate oxygen as a photosynthetic byproduct because they use hydrogen sulfide instead of water to convert solar energy into chemical energy (electrons).
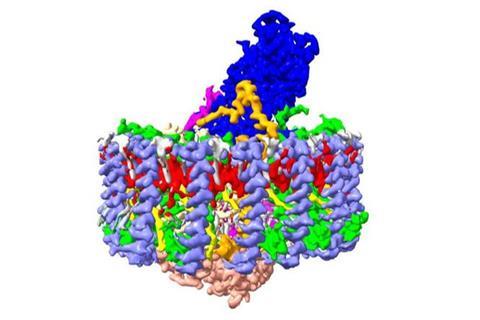
This process is orchestrated by a protein complex, the light-harvesting 1-reaction center (LH1-RC). Numerous PSB thrive in calcium-rich environments, such as hot springs and seawater.
In the three-dimensional LH1-RC structure, the LH1 antenna protein is typically associated with calcium. However, the photosynthetic mechanism remains elusive in Allochromatium vinosum, a model species of autotrophic bacteria capable of thriving in low-calcium or soft-water environments, as hypothetically, calcium is not involved in the photosynthetic process in this model.
Amino acid visualization
Using cryo-electron microscopy, researchers at the University of Tsukuba revealed the LH1-RC structures of this model species at a resolution that enabled individual amino acid visualization. These observations, published in Communications Biology, revealed calcium binding only at six specific sites in the LH1 subunit.
In contrast, the closely related thermophilic bacterium Thermochromatium tepidum displayed calcium attachment across all 16 LH1 subunits, indicating a calcium binding dependence on the amino acid sequence pattern. These results imply an evolutionary adaptation in this species, enabling it to bind trace amounts of calcium in low-calcium environments, thereby improving its thermal stability for photosynthesis.
These findings would potentially advance the efficient use of solar energy, and contribute to environmental protection, and highlight the capability of certain species to conduct photosynthesis in freshwater while detoxifying hydrogen sulfide, which is toxic to numerous organisms, into sulfur.







No comments yet