Particular families of Solute Binding Proteins recognise certain families of ligands, a finding that could help narrow down the search for the ligand that matches an uncharacterised SBP.
That in turn will make it easier to pinpoint valuable SBP-ligand pairings that can be used for biotechnological exploitations, such as enzyme evolution or biosensor construction.
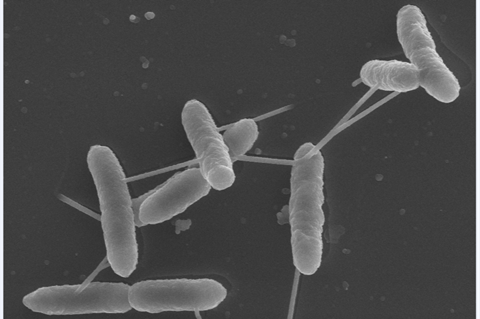
The discovery was made by researchers from the Estación Experimental del Zaidín in Granada, Spain, whose paper ‘Sensing preferences for prokaryotic solute binding protein families ’, was recently accepted by Microbial Biotechnology, an Applied Microbiology International publication.
Corresponding author Professor Tino Krell said they had found that the type of ligands recognized by Solute Binding Proteins (SBPs) is reflected in overall protein sequence similarities and have defined ligand profiles for many different SBP protein families.
Physiological importance
“Solute binding proteins are very abundant in microorganisms and are of central physiological relevance. SBPs present different solutes to transmembrane transporters to initiate compound uptake, activate different transmembrane signal transduction receptors and possess a number of additional, yet poorly understood functions,” he explained.
“SBPs are also of biotechnological relevance since they can serve as precursors for enzyme evolution and are used for biosensor construction. The action of SBPs is basically linked with any physiological feature of microorganisms, including mechanisms resulting in bacterial virulence.
“SBPs form a superfamily composed of at least 33 individual protein families (according to the Pfam database that uses sequence similarity as a means to classify proteins). The type of ligands recognized by SBPs is almost unlimited and includes for example amino- and organic acids, peptides, sugars, polyamines, quaternary amines, purines, metal ions, oxoanions, vitamins, quorum sensing signals and different siderophores.
“We wanted to find out whether the type of ligand recognized is reflected in overall SBP sequence similarity, or, in other words, whether members of a given SBP family bind specifically members of a given ligand family.”
Scouring the library
The team started by manually inspecting the totality of the three dimensional structures of SBPs deposited in the protein data bank and retrieved the ligands bound. Proteins were then classified according to their Pfam family and annotated with the corresponding ligand.
“This analysis showed that most SBP families recognized ligands that belong to a given ligand family. For example, we identified three families that each bound preferentially amino acids or sugars,” Professor Krell said.
“Other families were found to recognize preferentially purines, quaternary amines, iron and iron chelating compounds, oxoanions, bivalent metal ions or phosphates. This implies that ligand specificity is reflected in overall sequence similarity of SBPs.
“This is in contrast to the many sensor domains of bacterial receptors, for which ligand specificity is reflected in overall domain sequence similarity in only few cases.”
Just the right ligand
This find will help with one of the major challenges facing the team - that while SBPs can be easily identified using sequence analysis, the cognate ligands have only been determined experimentally for a small fraction of SBPs.
SBPs are of central physiological relevance and biotechnological relevance, and identifying the ligands recognized by SBPs is of crucial importance, not only for the basic research to understand bacterial physiology, but also for their biotechnological exploitation such as for enzyme evolution or biosensor construction, Professor Krell said.
“Our discovery that many SBP families are characterised by a specificity for ligands of a given family will permit us to rationally orient efforts to experimentally identify the ligand of an uncharacterised SBP,” he said.
“In other words, ligands of sensor proteins are frequently identified by high-throughput screening of compound libraries. Our discovery permits us to narrow down the compounds to be screened to a given family, which in turn will facilitate and accelerate the experimental identification of SBP ligands.”
This research was led by Prof. Tino Krell of the Estación Experimental dl Zaidín in Granada (Spain) and supported by grants from the Spanish Ministry for Science and Innovation, the Andalusian regional government Junta de Andalucía and the European Union.
‘Sensing preferences for prokaryotic solute binding protein families ’, was published by Microbial Biotechnology, an Applied Microbiology International publication.
Supporting documents
Click link to download and view these filesImage Pectobacterium artrosepticum
Image, Size 0.52 mb





No comments yet