When the COVID-19 pandemic first started, no effective anti-viral drugs were available to fight the disease. However, in record time, so-called monoclonal antibodies were developed as a lifesaving treatment.
Now, three years later, none of the approved antibodies work effectively against the new SARS-CoV-2 virus variants due to mutations that alter their spike protein.
While vaccines protect against severe disease, there is still an urgent need for effective virus-blocking agents for therapeutic or prophylactic use. This is particularly relevant for patients that are immunocompromised, meaning that they have a weakened immune system. Such patients can have a medical condition, or they are on immunosuppressive drug treatment. Examples are patients that suffer from severe primary immunodeficiency disease, recipients of organ or stem cell transplants or cancer treatment such as chimeric antigen receptor CAR-T-cell therapy.
Significant challenge
In response to this need, new monoclonal antibodies that neutralize recent virus variants by blocking their spike protein are being developed. However, again there is a risk that these agents may lose their efficacy when the virus continues to mutate and evolve, which poses a significant challenge to commercial development.
An alternative, attractive approach is to use soluble recombinant human angiotensin-converting enzyme 2 (ACE2) protein as a decoy receptor for SARS-CoV-2. The rationale is based on the fact that ACE2 is the cellular receptor for the virus, and thus, binding to ACE2 is a prerequisite for infection to occur.
So, while the virus may undergo mutations and thus evade recognition by spike-specific antibodies, it must maintain the ability to engage ACE2 in order to propagate. However, the challenge with soluble recombinant ACE2 is that it has a plasma half-life of only 10 hours in humans, which is not optimal when both systemic and mucosal tissue presence is needed.
Tailored ACE2 biologic
In the current work, the laboratory of Professor Jan Terje Andersen reports on a tailored ACE2 biologic, where ACE2 is fused to an engineered human albumin variant. Albumin engages a cellular receptor, FcRn.
Importantly, FcRn is broadly expressed, such as on mucosal epithelial cell barriers, and also on the endothelial cells that line the inside of blood vessels. Here, FcRn secures albumin a long plasma half-life as it rescues albumin from intracellular degradation. The designed albumin variant employed, binds strongly to human FcRn and secures that the ACE2 fusion proteins obtain an even longer plasma half-life.
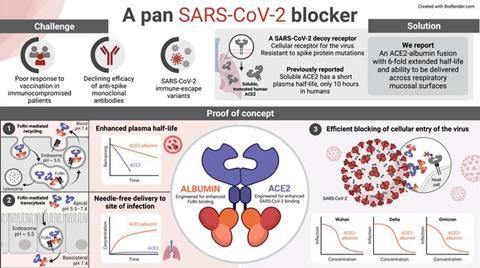
Furthermore, FcRn expressed on by the mucosal epithelial cells transports the fusion protein into the body in a non-invasive manner. The human ACE2 was also engineered for enhanced binding to the spike protein, and thus, the biologic showed potent ability to block cellular infection of all SARS-CoV-2 variants tested.
Potential for immunocompromised patients
“Insights into the complex biology of FcRn offers a range of opportunities in design of biologics that are tailored for improved pharmacokinetic properties as well as delivery across selective barriers of the body. The engineered long-acting and pan-SARS CoV-2 specific soluble ACE2 biologic is one such example that should be attractive to explore for prophylactic or therapeutic use among immunocompromised patients,” says Jan Terje Andersen, the study’s senior author.
The project was led by the Laboratory of Adaptive Immunity, headed by Professor Jan Terje Andersen. The laboratory is part of Precision Immunotherapy Alliance (PRIMA), a Norwegian Research Council Centre of Excellence, University of Oslo and Oslo University Hospital. It was carried out in collaboration with the MRC Laboratory of Molecular Biology, Norwegian Institute of Public Health, Norwegian University of Science and Technology, University of Tartu as well as University of Helsinki.
The study was mainly financed through the Research Council of Norway and the South-Eastern Norway Regional Health Authority.







No comments yet