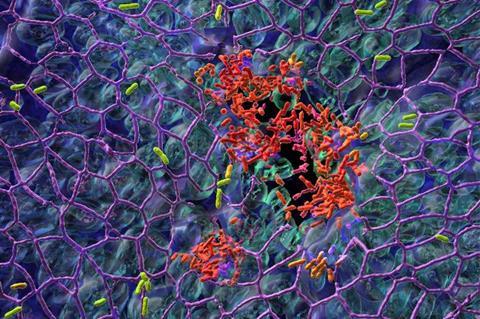
Researchers at the University of Basel have now uncovered why one of the most dangerous nosocomial pathogens is so difficult to combat. Pseudomonas aeruginosa follows a dual strategy, with some bacteria colonizing the tissue surface while others spread in the body.
The study provides important insights into the infection process and opens up new ways to treat infections.
Every year, about 6,000 patients die from nosocomial infections. One pathogen of major concern is Pseudomonas aeruginosa. This resistant bacterium colonizes the skin and mucosal surfaces and can cause life-threatening pneumonia particularly in immunocompromised patients.
Division of labour
The research team of Professor Urs Jenal at the Biozentrum, University of Basel, has now discovered why P. aeruginosa is successful in colonising surfaces: it practices a division of labour. While one fraction of the bacterial population adheres to the mucosal surface and forms a biofilm, the other subpopulation spreads to distant tissue sites.
Through this ’job sharing’ process, the pathogens increase their surface colonisation success. Protected in a biofilm, they can even resist antibiotic treatments.
In their study, recently published in Nature Microbiology, the scientists report on a stochastic genetic switch that is responsible for the division of labour and thus the lifestyle of bacteria – motile or sessile. After initial surface colonization, the pathogens do not simply divide at random, but rather form two functionally distinct subpopulations.
This behaviour is regulated by different levels of the bacterial signaling molecule c-di-GMP. Bacteria with high levels of c-di-GMP attach to surfaces and form a robust biofilm, while bacteria with low levels are motile and disperse into the surrounding tissue to colonise other parts of the host tissue. The concentration of c-di-GMP and consequently the behavioir of the bacteria is regulated by this stochastic switch.
’Stick and run’ mechanism
“The division-of-labour strategy enables the bacteria to rapidly respond to different stress conditions, because at any given time, a fraction of cells is optimally adapted to survive,” explains Dr. Christina Manner, first author of the study.
Bacteria in biofilm communities, for example, are protected from attacks by immune cells, while the fraction of dispersing motile bacteria can conquer new ground. This division of labour is also known as the ‘stick and run’ mechanism.
“We now better understand how Pseudomonas aeruginosa manages to spread and thrive on lung mucosa,” says the project leader Urs Jenal.
Achilles heel
“By identifying the genetic switch, we have tracked down the Achilles heel of the pathogen.”
The study not only provides valuable insights into the infection process but also reveals new therapeutic options to control infections with this dangerous nosocomial pathogen.
“We were also able to show that the recently discovered anti-biofilm compound Disperazol targets the same mechanism and flips the switch in favour of motile Pseudomonas cells, leading to biofilm dispersal,” adds Jenal. “This is a major step forward, as such agents open up new ways to eradicate difficult-to-treat biofilm infections.”




No comments yet