Scientists have designed a bacteria-targeted cargo system that is capable of inhibiting a quinolone resistant Escherichia coli biofilm using much lower levels of antibiotic.
The researchers, from Koç University School of Medicine in collaboration with College of Science, Department of Chemistry, encapsulated the antibiotic ciprofloxacin into superparamagnetic iron oxide nanoparticles (SPIONs) and conjugated the system to mannoside to target the bacterial fimbriae - hair-like protein polymers that are often linked to pathogenicity.
They are hopeful that the technique will offer a new design that will boost the efficiency of ciprofloxacin against bacteria that have become resistant to the antibiotic.
Anti-biofilm
The study, ‘Fimbria targeting Superparamagnetic Iron Oxide Nanoparticles enhance the antimicrobial and antibiofilm activity of ciprofloxacin against quinolone-resistant E.coli’ has been accepted by Microbial Biotechnology, an Applied Microbiology International publication.
“Among E.coli, quinolone resistance is highly prevalent, especially in countries where prescription rates of the antibiotic are high. Apart from quinolone resistance itself, multi-drug resistance is highly increasing which poses a worldwide threat as it opts out the last resort antibiotics for efficient treatment,” explained corresponding author Nazlı Ataç, a postdoctoral researcher at KUISCID (Koç University-İşbank Center for Infectious Diseases).
“To combat this threat, new designs are needed to increase the efficiencies of existing antibiotics and in this study, we aimed to increase the treatment efficiency of ciprofloxacin against ciprofloxacin resistant E.coli with a SPION based targeting system.”
Rapidly rising
”Deaths from antibiotic-resistant bacterial infections are expected to exceed the fatality rate due to cancer or other diseases in the next 50 years, with urinary tract infections being one of the problematic recurring infections that cause treatment failures,” she said.
E.coli is one of the most common bacteria causing urinary tract infections. The fimbria of E.coli can bind to host uroepithelial cells and form biofilm-like structures (intracellular bacterial communities).
Biofilms worsen the antibiotic treatment due to limited penetration of the antibiotic through the biofilm matrix. Biofilm related infections are one of the major concerns, especially in catheterized patients.
“Our goal was to overcome the penetration limitations and increase the ciprofloxacin efficiency even in the biofilm. With encapsulating ciprofloxacin to SPIONs with a mannoside tagging to further target the bacteria, we aimed to decrease the therapeutic dose of ciprofloxacin against ciprofloxacin resistant E.coli,” Dr Ataç said.
Delivery system
The team first synthesised superparamagnetic iron oxide nanoparticles (SPIONs), coated them with polyacrylic acid and conjugated them to mannoside to target the bacterial binding site (fimbria) and encapsulated ciprofloxacin into the design.
Synthesis of each group was followed by surface characterisations, as surface charge and size of the particles play a critical role in the appropriate interactions with target sites.
They found that SPION alone or mannoside-tagged SPIONs did not have an inhibitory effect against E.coli but when ciprofloxacin was encapsulated, they achieved 97.1 and 97.5% inhibition of two clinical E.coli isolates with a four-fold dose lower than ciprofloxacin MIC of the isolates.
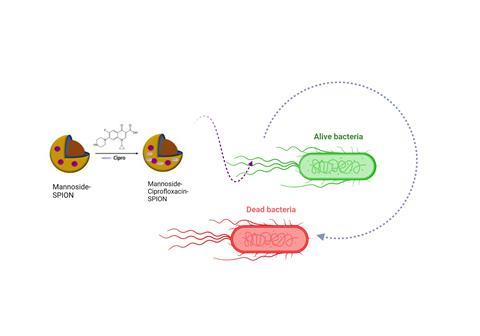
“Besides, biofilms were inhibited by 86.9% and 98.5%, respectively, with the same low dose. These promising results demonstrated the potent enhancement of ciprofloxacin efficiency with fimbria targeting of SPION cargo system, considering further use of the design in alternative and/or complementary therapy approaches of urinary tract infections,” Dr Ataç said.
Surprising find
What surprised them was how effective the anti-biofilm activity proved to be along with the planktonic inhibition, using a lower dose of ciprofloxacin - bearing in mind the very high doses that are usually required to eliminate biofilms (up to 1000x higher doses).
“Our design seems to be promising to test such cargo systems against different types of bacteria,” Dr Ataç said.
“Various modifications of the target site with respect to the specific features of the bacteria of interest brings the advantage to improve the activity of existing antibiotics and this may help to finding new ways to treat bacterial infections.”
The study was conducted as an in vitro study, and to further relate its biocompatibility and clinical applicability of the cargo delivery system as an alternative drug, the next step will be to proceed to in vivo studies.
Multi-disciplinary
The multi-disciplinary study at Koç University School of Medicine was conducted in collaboration with the College of Science, Department of Chemistry.
Synthesis and characterisation steps of SPION cargo design were supervised by the Prof. Funda Yağcı Acar team, Polymer and Nanomaterials Research Lab. Antibacterial and anti-biofilm activity tests of the SPION system against ciprofloxacin resistant E.coli were supervised by the Prof. Füsun Can team, Medical Microbiology Lab / KUISCID (Koç University-İşbank Center for Infectious Diseases).
The study was funded by the Scientific and Technological Research Council of Türkiye (TÜBİTAK) with the project number 118S547.
‘Fimbria targeting Superparamagnetic Iron Oxide Nanoparticles enhance the antimicrobial and antibiofilm activity of ciprofloxacin against quinolone-resistant E.coli’ appears in Microbial Biotechnology.
Topics
- Antimicrobial Resistance
- Applied Microbiology International
- Bacteria
- Biofilms
- ciprofloxacin
- Community
- Escherichia coli
- fimbria
- Funda Yağcı Acar
- Füsun Can
- Innovation News
- Koç University School of Medicine
- Koç University-İşbank Center for Infectious Diseases (KUISCID )
- Kübra Onbaşlı
- mannoside
- Middle East & Africa
- Nazlı Ataç
- One Health
- Pharmaceutical Microbiology
- quinolone-resistant
- SPIONs




No comments yet