Men and women show different responses to COVID-19, with men having a higher risk from infection. The underlying cellular basis for this difference is not fully understood, but now a research group from Osaka University have uncovered sex-specific differences in a type of immune cell called regulatory T cells, or “Treg cells”, and in the production of antibodies, as part of the response to COVID-19 infection.

The study appears in Proceedings of the National Academy of Sciences.
The immune system is responsible for clearing viral infections, as well as producing many of the acute symptoms of COVID-19, and it is critical to understand the changes that occur to the immune system of infected patients. The “humeral response”, the production of antibodies, is dysregulated in COVID-19. Cells called “Treg cells” were suspected to be responsible for this, as their role in the immune system is to regulate other immune cells and suppress their activities to control the strength of the immune response.
Circulating Tfr cells
Of particular relevance are “Tfr cells”, a subset of the Treg cell population responsible for control of antibody production. The team observed that male patients lose circulating Tfr cells at a faster rate than female patients, and identified sex-specific differences to a whole network of different cell types that are associated with the production of antibodies.
Many COVID patients begin to produce “autoantibodies” as part of their response to the virus. These antibodies are aimed at proteins produced by the human body instead of targeting the virus and can neutralize protective host factors, and the production of these may play a critical role in how the infection progresses.
“Regulation of the immune system by Treg cells may therefore be key in understanding susceptibility to, and recovery from, COVID-19,” explains first author Jonas Nørskov Søndergaard.
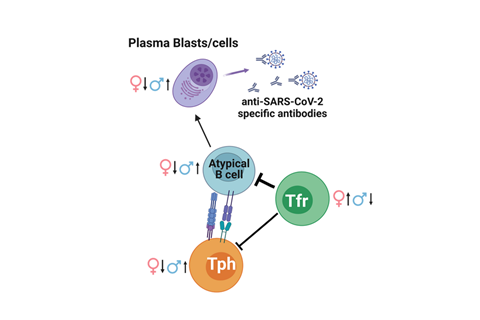
The team used an approach known as single-cell proteomics by mass spectrometry, allowing individual immune cells to be identified and analyzed. This showed that patients with COVID-19 have changes to the ratio between circulating Tfr cells and a network of other cells associated with the production of antibodies, which in turn is strongly correlated with the antibody levels.
A sex bias was seen in this response, with females having more circulating Tfr cells while males had higher antibody levels.
“This provides significant cellular evidence of dysregulated antibody responses in COVID-19 patients,” explains senior author James Badger Wing. “The reduction of cTfr observed in all COVID-19 patients, but particularly in males, may underlie this dysregulated antibody production.”







No comments yet