Future medicines will probably be made up of a cocktail of compounds that inhibit different bacterial targets, including some that act against their motility and chemotaxis mechanisms, a new review suggests.
The paper, ‘Targeting motility and chemotaxis as a strategy to combat bacterial pathogens’, by scientists at CSIC in Spain, has been accepted by Microbial Biotechnology, an Applied Microbiology International publication.
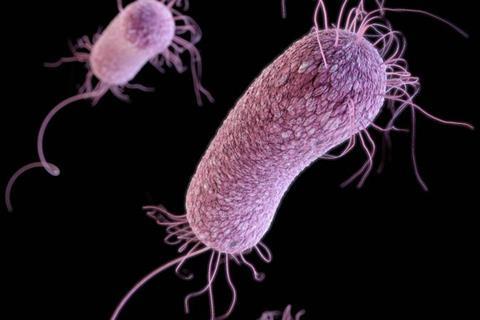
Large numbers of human and plant pathogens require motility and chemotaxis (directed, flagellum-based motility in signal gradients) for efficient virulence, and in many cases chemotaxis is required for the initial stages of infection - the entry into the host and the attachment of the pathogen to host tissues.
Alternative strategy
“In this article we have discussed the question whether the inhibition of motility and chemotaxis is an alternative (to antibiotics) strategy to fight pathogens,” explained corresponding author Professor Tino Krell.
“The increasing number of antibiotic-resistant bacterial strains is among the global challenges of mankind that underlines not only the necessity to develop new antibiotics and pesticides, but also to identify alternative antimicrobial strategies.
“Antibiotics, our main weapons to fight bacterial pathogens, either kill or slow down bacterial growth, resulting in an evolutionary pressure that favours the appearance of further antibiotic-resistant bacteria.
Molecular mechanisms
“As an alternative, anti-infective therapy is based on targeting molecular mechanisms that lead to disease but that do not interfere with bacterial growth. One such alternative mechanisms may be targeting the motility and chemotaxis of pathogens.”
The authors begin by summarising articles that report a reduction in motility and chemotaxis using different compounds.
“The chemotactic mechanism is highly complex, offering a large number of targets for inhibition. Data have shown that chemotaxis can be inhibited by a number of different mechanism such as for example by interfering wIth signal recognition or by various ways to inhibit flagellar rotation,” Professor Krell said. “In a significant number of studies, the reduction in chemotaxis was shown to induce a reduction in virulence.”
Pros and cons
The authors then go on to consider the advantages and disadvantages of an anti-infective strategy based on chemotaxis inhibition.
Major advantages of this strategy are that the proof-of-concept has been obtained and that compounds with IC50 values (half maximal activity) of and below 10 μM are available. Furthermore, this strategy does not kill or slow down bacterial growth, thus reducing the possibility of the generation of resistant strains.
“Major disadvantages are that not all pathogens perform chemotaxis, and that in a number of pathogens the suppression of chemotaxis reduces, but does not abolish virulence,” Professor Krell said.
Future medicines
“Future medicines are likely cocktails of compounds that inhibit different bacterial targets. We finish with the conclusion that, considering the experimental data available as well as the advantages and disadvantages, the interference with bacterial motility and chemotaxis is a reasonable strategy to fight pathogens.
“Further research is necessary to increase the efficiency of existing chemotaxis/motility inhibitors. These inhibitors target only some of the multiple possible target proteins of the chemotactic apparatus, whereas other targets are still unexplored. Further research is necessary to explore the effect of interfering with the activity of these unexplored targets.”
The study was led by Prof. Tino Krell from the Estación Experimental del Zaidín in Granada, Spain.
‘Targeting motility and chemotaxis as a strategy to combat bacterial pathogens’ appears in Microbial Biotechnology.





No comments yet