A first-of-its-kind study led by Duke-NUS Medical School has shown that prior maternal immunity to the dengue virus substantially increases the risk of severe birth defects induced by Zika virus infection during pregnancy.
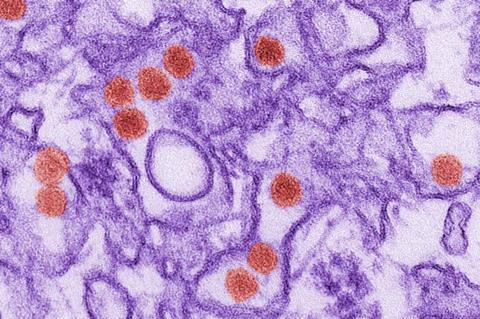
The research, published in Science Translational Medicine, found that pre-existing dengue antibodies in pregnant mothers lead to severe microcephaly and brain damage in the foetus after Zika infection compared to Zika-infected foetuses from dengue antibody-naïve mothers. Microcephaly is a birth defect where a baby’s head is smaller than expected and is one of the hallmark features of congenital Zika syndrome.
“This study provides compelling evidence that a mother’s immune history with dengue virus can put her baby at greater risk if she contracts Zika virus during pregnancy,” said senior author Associate Professor Ashley St John, from Duke-NUS’s Emerging Infectious Diseases (EID) Programme. “It highlights a complicated interplay between these two closely related viruses and may provide new insights into why some foetuses experience much more severe outcomes from Zika infection than others.”
Modelling dengue exposure
The team first infected a preclinical model with dengue virus to model dengue exposure in humans. After resolving the infection, the dengue-immune females were bred and infected with Zika virus early during gestation, alongside a control group that were infected during pregnancy with Zika virus without previous dengue virus exposure.
They found that the foetuses from dengue-immune mothers infected with Zika virus showed severe microcephaly, with head circumferences around 40 per cent smaller compared to healthy foetuses throughout pregnancy. Brain tissues of these foetuses also showed more abnormalities, such as a thinner cerebral cortex, destruction of neurons, bleeding, inflammation and calcium build-up - all hallmarks of severe Zika-related damage.
“Seeing multiple cases of severe microcephaly, brain damage and even foetal demise in this preclinical model really highlights how prior dengue immunity can aggravate congenital Zika syndrome,” said first author Dr Wilfried Saron, Research Fellow with Assoc Prof St John’s lab in the EID Programme.
Perfect storm
The findings may explain why Zika infection outcomes in human pregnancy seem to vary dramatically, with some resulting in foetal demise or severe microcephaly while others experience mild or no birth defects.
“In regions where dengue virus is widespread, such as Brazil, we believe that pre-existing dengue immunity could create a ‘perfect storm’ that worsens damage to the foetus when Zika infection occurs during pregnancy,” said Assoc Prof St John.
“However, our data suggest that prior dengue immunity does not make Zika infection or symptoms worse for the mother - so the driver of severe foetal damage is likely to be the unique features of the immune environment in utero, like the mother’s antibodies.”
Managing risk
The results have major implications for understanding the viral and immune factors that influence congenital Zika syndrome, which is important for managing risk.
The findings also suggest that treating pregnant women who have dengue antibodies against Zika-induced immunopathology could be beneficial.
“This study raises the possibility that we could reduce Zika-related birth defects by developing approaches that target detrimental maternal antibodies from reaching the foetus,” said Dr Saron.
Next steps
Moving forward, the researchers will continue using their preclinical pregnancy model to unravel how exactly prior dengue immunity exacerbates foetal damage upon Zika infection, and develop and test treatments that could prevent severe birth defects. Targeting FcRn, which is a receptor that can translocate antibodies from the mother to the foetus, is one promising approach that they aim to develop.
“Our ultimate goal is to find ways to protect foetuses from Zika and related viruses, even in mothers with prior flavivirus immunity. The development of dengue vaccines also amplifies the urgency of this work,” said Assoc Prof St John.
“This collaborative research elegantly unravels the complex interplay between dengue and Zika virus infections during pregnancy in a preclinical model, enabling the team to uncover new insights into the pathogenesis of congenital Zika syndrome. It’s also timely because several dengue vaccines are currently under development or in use, and it will be crucial to determine whether they could have unintended consequences if Zika emerges again,” said Professor Patrick Tan, Senior Vice-Dean for Research at Duke-NUS.



No comments yet