An international study led by MELIS-UPF researchers from the Infection Biology and Molecular Virology laboratories has identified and characterized Schlafen 12 (SLFN 12) as a novel HIV restriction factor.
SLFN 12 shuts down viral protein production and helps virus-infected cells to escape from anti-HIV therapy and immune responses, and the findings pave the way for improving therapeutic strategies that aim to cure HIV infections.
Human Immunodeficiency Virus (HIV) infections, if left untreated, lead to the gradual destruction of the immune system, AIDS, in its final stages. Worldwide, some 650,000 individuals die of AIDS each year, making it a major threat to human health. Nonetheless, despite there being no general cure for an established infection, appropriate antiretroviral therapy enables people living with HIV to lead a relatively healthy life. Unfortunately, once treatment is stopped, the virus returns from a reservoir of latently infected cells.
Major barrier
“Latency is a major barrier impairing virus elimination in HIV-infected individuals. We will not be able to cure an existing infection until we will get rid of latently infected cells. This is why it is essential to understand how latency works,” explains Andreas Meyerhans, ICREA research professor at UPF who has coordinated the study together with Juana Díez.
The paper, published in Communication Biology, has identified and characterized SLFN12, a protein that restricts the production of viral proteins by cleaving specific cellular tRNAs, the building blocks for protein construction. As a consequence, in the presence of active SLFN12, HIV-infected CD4 T cells are not able to complete the virus production process but keep its templates, HIV RNA, in a latent state.
“SLFN12 impairs protein production, restricting the production of viral particles. Such cells are latently infected, invisible to the immune system and anti-HIV therapies,” says Mie Kobayashi-Ishihara, first author of the manuscript.
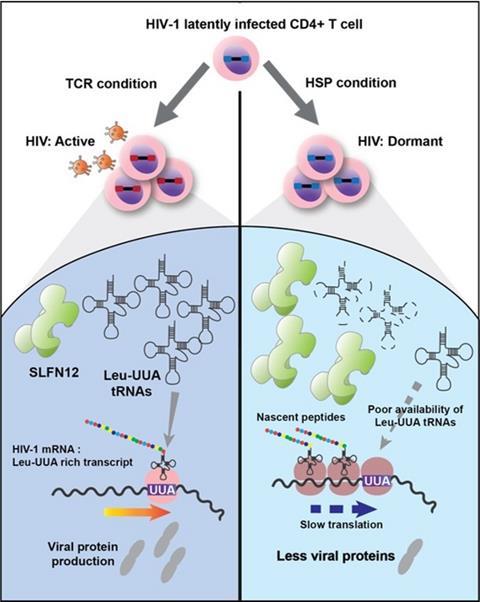
The study also reveals how SLFN12 can specifically inhibit HIV protein production without blocking cellular protein production. “SLFN12 cleaves Leucine-UUA tRNA, a building block that is rarely used for cellular proteins but essential for HIV viral proteins,” Juana Díez explains.
This finding opens the possibility of designing new therapeutic strategies against HIV.
“Blocking SLFN12 antiviral functions should increase viral protein expression and thus, enable the host immune system and antiviral drugs to better eliminate viral reservoirs. Once you start producing the virus, it becomes visible again. You get your target back. So, you can attack it, and hopefully, definitely eliminate the latent infected cells,” concludes Meyerhans.
Schlafen is a German word meaning “sleeping” that gives its name to a family of proteins involved in cancer, drug sensitivity and antiviral functions.







No comments yet