Scientists have unravelled part of the Coenzyme Q10 biosynthetic pathway in Agrobacterium tumefaciens, paving the way to enhancing the bacterium so that it can produce the probiotic on an industrial scale.
The study exploring the pathway, ‘Combining in silico and in vitro approaches to understand the involvement of MEP and shikimate pathway in Agrobacterium tumefaciens for enhanced CoQ10 production’, has been accepted for publication by the Journal of Applied Microbiology.
The team, based at the Regional Centre for Biotechnology and the Central University of Haryana, used a systems and synthetic biology approach to clarify the CoQ10 biosynthetic pathway in Agrobacterium tumefaciens, explained the corresponding authors.
“In silico and in vivo analysis displayed partial completeness of the CoQ10 metabolic biosynthesis pathway, ” they said.
“The study highlighted the importance of characterizing unknown genes in the CoQ10 pathway. Overall, the study’s findings will further efforts to metabolically engineer Agrobacterium species for increased CoQ10 synthesis.”
Global market
The market for Coenzyme Q10 has been expanding quickly, with a market size of over $890 million in 2017 in the last five years.
CoQ10 has been widely used as a dietary supplement, as well as for nutraceuticals, immune booster and in cosmetics. It is also widely employed for therapeutic applications such as heart disease, Alzheimer’s and Parkinson’s diseases, and breast cancer.
It is an important molecule involved in cellular energy production and is also known for its antioxidant role as it protects the body against oxidative stress.
However, the synthesis of CoQ10 in the body declines with age, resulting in the need for external supplementation.
Undiscovered pathway
Its biological production using microbial strains has been hampered due to inadequate pathway understanding, so this study aims to unravel a previously undiscovered pathway, opening avenues to enhance CoQ10 production.
“Currently, CoQ10 is produced on an industrial scale through the fermentation by some natural or mutant microorganisms such as Agrobacterium tumefaciens, Paracoccus denitrificans, Rhodobacter sphaeroides or by recombinant Escherichia coli. But the yield of CoQ10 is far from industrial realisation,” the corresponding authors said.
“A variety of microbes naturally produce CoQ10, making it difficult to select a model organism. We performed pathway analysis which indicated A. tumefaciens as the most suitable host for our study.
“However, there exist gaps in the current understanding of the pathway. Using various systems and synthetic biology tools, we modelled the CoQ10 biosynthetic pathway. The pathway can be exploited for enhancement of CoQ10 using Agrobacterium sp.”
Microbial bioproduction
Several microorganisms that naturally produce this molecule are used for commercial production of CoQ10. Microbial bioproduction is the most viable approach for the large-scale synthesis of CoQ10. However, the current CoQ10 yields do not meet market demands.
“Several reasons can be assigned for suboptimal commercial production of CoQ10 including the choice of model organism, culture conditions and cost of production,” said the corresponding authors. “Moreover, the metabolic pathway leading to CoQ10 biosynthesis has not been completely elucidated in many industry-friendly microbes.
“This study was designed to understand CoQ10 metabolic pathway and increase the yields thereafter.
“We explored the CoQ10 biosynthetic pathway in 10 microbes. The prokaryotic and eukaryotic organisms were scanned to obtain the in-depth knowledge about the synthesis of CoQ10 metabolic network. The important contribution of regulatory pathways was also examined.
Key genes
“Through our analysis, we found that the biochemical pathway leading to CoQ10 formation in Agrobacterium sp. is not fully characterised and a few key genes from the metabolic network are not annotated yet.
“We identified the previously unknown homologs of these enzymes in Agrobacterium sp. The biological roles of these genes can be investigated further to metabolically engineer Agrobacterium sp to enhance CoQ10 production.”
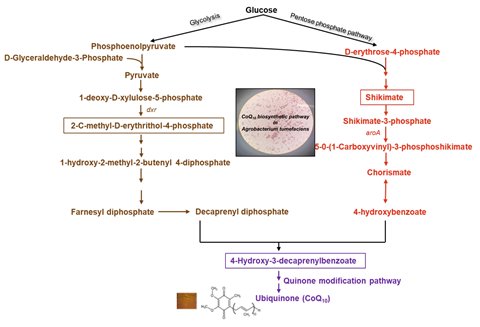
The authors said this study contributes to our knowledge of the CoQ10 biosynthetic pathway and may help to develop future metabolic engineering strategies leading to increased CoQ10 production in A. tumefaciens.
“We also identified the earlier uncharacterized homolog of the ubiC, ubiI and ubiF gene encoding a putative hydroxylase. These enzymes are involved in the bioproduction of CoQ10. Additional studies are needed to characterise these genes,” they said.
This study is supported by an Institutional Research grant.
‘Combining in silico and in vitro approaches to understand the involvement of MEP and shikimate pathway in Agrobacterium tumefaciens for enhanced CoQ10 production’ appears in the Journal of Applied Microbiology.
Topics
- Agrobacterium tumefaciens
- Applied Microbiology International
- Asia & Oceania
- Central University of Haryana
- Coenzyme Q10
- Healthy Land
- Industrial Microbiology
- Journal of Applied Microbiology
- Microbial Biotechnology
- One Health
- Paracoccus denitrificans
- Pharmaceutical Microbiology
- recombinant Escherichia coli
- Regional Centre for Biotechnology
- Rhodobacter sphaeroides
- Synthetic Biology
- Systems Biology







No comments yet