When infants are born, they are immunologically prepared to survive in a microbial world. Foetal immune development is important for infant and lifelong health, but our understanding of the process is vastly incomplete. Specifically, it is not agreed upon when a healthy foetus is first exposed to microbes, and whether these microbes are alive and colonize, or if we are just talking about microbial components and metabolites.

It was more than a hundred years ago that scientists, with the help of bacterial culture method and microscopy, established that the foetus was sterile. This notion was further cemented after the 1960s when scientists were able to derive germ-free mammals through caesarean sections.
It was only after 2010 that a group of scientists applied novel next-generation sequencing to samples collected from the foetal environment, such as the placenta and amniotic fluid, and claimed the discovery of entire constellation of microbes.
Suggestions were made that these microbes were alive and represented microbiomes (interactive communities that not only contain bacteria but also microbes such as fungi and viruses) that are part of a healthy pregnancy, seed the foetus, and aid its development. Soon, the notion of a foetal microbiome spurred an entire research field that went beyond the characterization of microbial communities in utero and attempted to characterize their medical importance for pregnancy outcomes, infant health, and the development of diseases later in life. The result were hundreds of publications, targeted funding initiatives, and tens of millions of euros/dollars spent.
Not everyone in the scientific community, however, shared the excitement. When the first papers supporting the “in utero colonization hypothesis” were published, I was very surprised as I considered the historic evidence that had established the sterile womb paradigm (100 years of culture studies and derivation of germ-free mammals) much stronger than the sequencing data.
READ MORE: The usually sterile womb
Sequencing approaches only detect DNA and can therefore not establish viable colonization. In addition, counter arguments were brought forward by sceptics that a foetal microbiome would violate fundamental concepts in immunology and reproductive biology.
For example, the foetal immune system would likely be incapable of controlling the bacteria that were detected (many of which are opportunistic pathogens). For this specific reason, the placenta is full of immunological and anatomical features to prevent microbes from crossing or colonizing. When the field took off, I was surprised at how easily the advocates of the foetal microbiome would dismiss these fundamental concepts.
This scepticism was then supported by an increasing number of studies that showed that the microbial signals detected in the foetal environment were due to contamination during sampling, DNA extraction (the so-called kitome), or DNA sequencing (the splashome).
Opposing conclusions
A lively and sometimes contentious scientific debate arose with views that were pretty much irreconcilable. The controversy culminated when four studies published in high-tier scientific journals (see references below) that characterized the microbiology of the human foetus directly came to completely opposite conclusions. Two found viable microbial populations in human foetal intestines and organs and implicated their presence in foetal immune development. The other two reported no microorganisms in the foetal meconium and intestines.
Such disagreements on a fundamental issue, and the uncertainties that they cause, pose a substantial challenge for scientific progress. They divert finite resources into research that results in no advancement for foetal and maternal health as well as risking misguided attempts to therapeutically modify microbiomes that don’t exist. The negative impact on scientific credibility, and the public’s trust in science, is also not negligible.
It is not a great message to the lay public that microbiome experts can’t even agree on just the sheer existence of entire microbiomes at a particular location despite significant public-funded research. Such messages could have lasting effects on the public trust in the scientific process, especially since the scientific self-correction process is now much slower than the transfer of information through social media.
Team of experts unanimously refuted concept of foetal microbiome
To resolve the debate, we assembled a team of 46 leading experts to assess the evidence and review all aspects of the contradictory data, focusing on the recent publications on the microbial analyses of human foetuses.
Given the controversy, our aim was to approach the problem from different angles and to include experts from a wide variety of disciplines, ranging from reproductive biology and obstetrics over microbiology and bioinformatics to immunology. We were further interested in a balanced approach, and therefore involved a majority of authors with no previous involvement in the debate. With 46 experts, this can generate multiple caveats and qualifiers, but it is noteworthy that none of the authors were equivocal in their conclusion.
In our publication in the journal Nature, the team unanimously refutes the concept of a foetal microbiome. We conclude that the detection of microbes in foetal tissues was due to contamination of samples drawn from the womb, or that occurred during sequencing and DNA extraction. Experts further concluded that the existence of live microbial populations in healthy foetal tissues is not compatible with fundamental concepts of reproductive biology, immunology, clinical microbiology, and the ability to derive germ-free mammals. Bacterial colonization does happen occasionally in human foetuses but is always linked to pathology, and thus cannot be considered a microbiome.
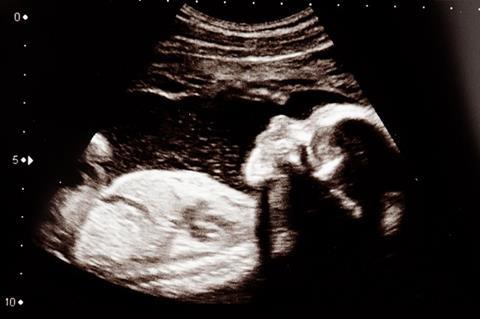
We are hopeful that this consensus provides guidance for the field to move forward in that it establishes a solid scientific foundation to concentrate research efforts where they will be most effective. It remains imperative to elucidate the mechanisms responsible for the maturation of the foetal immune system.
However, knowing that the foetus is sterile does allow scientists to adjust their methodology and emphasize studies on microbial metabolites and components crossing the placenta and not life microbes. Our conclusions further emphasize that colonization by bacteria happens during birth and in early post-natal life, which is where therapeutic efforts on modulation of the microbiome to improve infant and lifelong health should be focused. I am indebted to all the crucial contributions made by all authors of this publication.
Cautionary tale
Our team also provides guidance on how scientists can avoid pitfalls of contamination in the analysis of other low-biomass samples, meaning samples where microbes are expected to be absent or present at low levels, such as internal organs and tissues.
The pursuit of a foetal microbiome serves as a cautionary example of the challenges of sequence-based microbial analyses and emphasizes the need for a trans-disciplinary approach that goes beyond contamination controls by also incorporating biological, ecological and mechanistic concepts.






No comments yet