Improving biosecurity in food production is a primary concern for consumers and government food safety agencies. The chicken gut possesses a diverse and complex microbiota that plays a vital role in the digestion and absorption of nutrients, as well as in immune system development, vitamin and amino acid production and prevention of pathogen colonisation.
Poultry meat is widely consumed and demand increases with population growth. Thus, providing sustainable, safe chickens is critical for the growing human population. The microbiome of chickens contains bacteria that can be pathogenic to humans, and indeed potentially to the chickens themselves. Campylobacter are mainly spiral-shaped, fastidious bacteria growing in microaerobic conditions. Currently, there are over 30 species of Campylobacter and 10 subspecies, with C. jejuni and C. coli being the main causes of human infection. Campylobacter is the leading cause of bacterial foodborne gastroenteritis within humans worldwide with an estimated 550 million cases per year (including 220 million children under the age of 5 years). The economic burden of Campylobacter infection is severe, costing the UK government alone over £1 billion per annum. Campylobacter is a zoonotic pathogen, which means it can spread from animals to humans. Typically, this is via contaminated food, especially raw or uncooked poultry. Additional sources exist and include contact with animals and their environment, contaminated water, raw milk and eating contaminated fresh produce. International travel to low-resource countries can also increase the risk of infection.
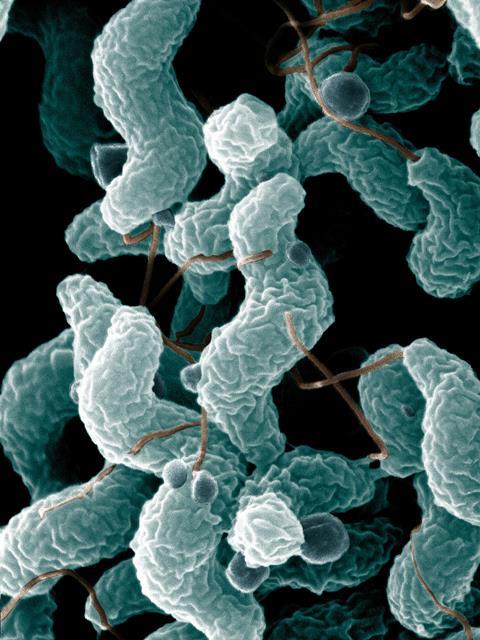
The increasing frequency of infection and the rise of antimicrobial resistance mean that development of new interventions to reduce Campylobacter in the food chain is a global necessity. Thus, the question remains, can investigating the chicken gut microbiome provide us with novel intervention and control strategies against Campylobacter?
Changes in the gut microbiota over time
To investigate the drivers of microbial change in the chicken gut over time, and how this relates to Campylobacter prevalence within a natural setting, Assistant Professor Ozan Gundogdu performed the first day-to-day microbiome analysis of the chicken caecum from day 3 to 35, with metadata such as chicken weight and feed conversion rates also included.
In this study, 16S ribosomal RNA (rRNA) short-read metagenomic sequencing was performed using Illumina technology. Variation was observed with rapidly increased microbial diversity up to day 12, creating an opportunity for Campylobacter appearance at day 16 before stabilisation of the microbial diversity after day 20. Microbial variation over time was most likely influenced by the diet of the chickens whereby significant shifts in genera were detected upon changes in feed regimen. However, this previous study was only performed for the caeca, and gives an incomplete picture of the chicken gut microbiome.
Applying bacterial 16S gene sequencing allows us to investigate the composition and diversity of a microbial community. However, there are limitations in relation to the sensitivity of the method. Long-read shotgun metagenomic sequencing is an effective way to enable identification down to species level, and allows us to perform classic environmental community metric analyses. Therefore, to obtain an improved understanding of the chicken gut microbiome and Campylobacter prevalence, the aim of my research is to perform a comparative, longitudinal, day-to-day study between the chicken caeca and additional organs (the duodenum, small and large intestine) using long-read shotgun metagenomic sequencing. Long-read shotgun metagenomics involves randomly fragmenting DNA into smaller segments, sequencing them, then stitching the DNA together using bioinformatics to identify the bacteria present in each sample.

Natural products and Campylobacter
My second aim focuses on how to use natural antimicrobials against Campylobacter. Typically, antibiotics have been widely utilised as growth performance enhancers in poultry. However, it is well known that antibiotic use can lead to the generation of antibiotic-resistant, pathogenic bacteria. Therefore, the routine supplementation of antibiotics into animal feed was banned as a method of growth promotion in 2006 by the European Union (EU). This creates a necessity to develop alternative methods to improve performance and potentially reduce numbers of pathogenic bacteria. A vast range of strategies currently exist, including hygiene and biosecurity farming practices, chemical feed additives, bacteriophage application, vaccination, passive immunisation, competitive exclusion cultures, host genetic selection strategies, bacteriophage therapy and bacteriocin application. Despite their application, however, there has been little progress in the reduction of Campylobacter in the food chain.
Here, I aim to investigate the impact of different natural antimicrobials on the growth of Campylobacter as a method to reduce these bacteria in the food chain. Antimicrobials are also important to control multidrug-resistant strains of Campylobacter that are rapidly developing. As an example, the efficacy of Auranta 3001 (a mixture of organic acids and plant extracts) in reducing the ability of C. jejuni and C. coli to invade epithelial cells in vitro was investigated. It was indicated that the antimicrobial agent has a direct effect on C. jejuni and C. coli and caused a significant reduction in invasiveness. A previous study from the Gundogdu group monitored the impact of Omega-3 and different industrial system parameters on the chicken microbiome and Campylobacter. Three different production systems, ‘Normal’, ‘Higher Welfare’ and ‘Omega-3 Higher Welfare’, were investigated in an industrial farm setting at day 7 and day 30, with a range of extrinsic parameters correlating performance with microbial dynamics and Campylobacter presence. It was found that use of Omega-3 provided a diet-specific microbiome that favoured the growth of the chicken.

Arrowroot prebiotics
Natural antimicrobials have been used as a viable alternative for pathogen reduction. I will be evaluating the effect of arrowroot extract (Maranta arundinacea) on the growth performance of Campylobacter by growing different strains at 37°C and 42°C to mimic human and avian body temperatures. Arrowroot is a tropical plant that produces an edible starchy tuber. Historically, it has been used for its medicinal properties.

The arrowroot plant is native to South America and to the West Indies, where the plant is used as a dietary staple. Arrowroot powder has also been used in wound healing. Preliminary data suggest that there are two kinds of active substances in arrowroot extracts: (i) a starch; and (ii) a protein. Previous studies have shown that the starch-rich arrowroot contains prebiotic fructo-oligosaccharides, which can be used to obtain a higher biomass of probiotics. Prebiotics are compounds that induce the growth of certain bacteria. Prebiotics were defined in 1995 as ‘non-digestible food ingredients’, such as fructo-oligosaccharides that have a beneficial effect on the host by selectively stimulating the growth of pre-existing bacterial species and/or activity in the colon. In general, prebiotics appear to be effective in poultry and reduce colonisation of foodborne pathogens such as Salmonella in the gastrointestinal tract (GIT). Therefore, my project will aim to understand the effectiveness of prebiotics from the arrowroot plant to limit Campylobacter growth in poultry. Furthermore, the antioxidant and anti-inflammatory activity of the phenolic compounds of arrowroot may be studied and developed for potential use as a control strategy against Campylobacter.

Overall, my main objective is to understand the mechanisms supporting the role of natural antimicrobials. I will look at concentration and time of administration in relation to performance efficiency and pathogen reduction. By doing so, I will provide a practical outcome that benefits multiple stakeholders, including those in academia, agriculture, policymakers, and the public.
https://pixabay.com/users/kriemer-932379/









No comments yet