This study, published in Science China Life Sciences, is reported by Rong Li’s group from the Peking university third hospital. Recurrent implantation failure (RIF) presents a significant challenge in the realm of assisted reproductive technologies, leading to considerable psychological stress and financial burdens for individuals experiencing infertility. The underlying mechanisms contributing to RIF remain poorly understood, highlighting the necessity for comprehensive research in this area to inform clinical practices.

Recent investigations have pointed towards an imbalance in reproductive tract microbiota as a potential factor associated with RIF, offering a novel and promising avenue for exploration. The team has revealed noteworthy findings regarding the endometrial microbiota during the secretory phase. Notably, the authors observed significant differences in the uterine microbiota between the RIF group and the control group.
Further comparisons of endometrial tissue metabolites during the secretory phase indicated that 34 metabolites were significantly elevated, whereas 19 metabolites, including dimethylglycine (DMG), were significantly reduced in the RIF group. Correlation analyses revealed meaningful relationships between the differentially abundant microbiota and the identified metabolites within the endometrium, suggesting intricate interactions at play.
Endometrial microbiota
To delve deeper into these findings, the authors conducted an experiment involving the transplantation of endometrial microbiota (EnM) from RIF patients into the uterine cavity of Sprague-Dawley (SD) rats.
This intervention resulted in a significant alteration of the uterine microecological environment, as evidenced by decreased levels of Hoxa-10 and Lif, along with a reduction in embryonic implantation sites.
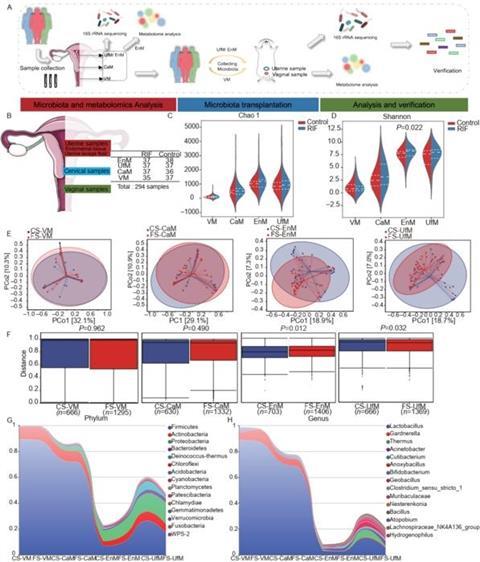
Investigating the underlying mechanisms, the authors found that microbiota transplantation led to a decrease in the proportion of regulatory T cells (Treg) within the uterus and a corresponding reduction in DMG expression.
Potential therapies
Importantly, the authors further demonstrated that supplementation with DMG might have the potential to counteract the decline in Treg cells and the associated impairment of endometrial receptivity stemming from disturbances in the endometrial microbiota. Thus, the alterations observed in the EnM of patients with RIF might have far-reaching consequences, leading to changes in endometrial metabolites, decreased Treg-cell proportions, compromised endometrial receptivity, and ultimately, recurrent implantation failure.
In conclusion, this study emphasizes the critical role of endometrial microbiota in the context of RIF and underscores the importance of further research to elucidate the complex interactions between microbiota, metabolites, and reproductive success. Addressing these factors could pave the way for novel therapeutic strategies aimed at improving outcomes for individuals facing recurrent implantation failure.







No comments yet