Researchers from the Royal Veterinary College (RVC) and University of Oxford have led an innovative project investigating the progression of malaria infection and the role of the parasite to better aid the development of an effective malaria vaccine and significantly reduce rates of deaths from the disease.
Malaria is a deadly disease caused by the Plasmodium parasite which infects more than 200 million people a year, causing approximately 600,000 deaths annually worldwide with young children in Africa disproportionately affected. With numbers of infections expected to rise in the coming years due to growing parasite resistance to frontline anti-malarial drugs, there is a significant need for the development of new malaria vaccines.
The study, which was funded by the Wellcome Trust, observed the cycles of parasites invading red blood cells (RBCs), replicating within them, and then spreading to infect new RBCs. From its observations, the project has determined that during the invasion of new RBCs by the parasite, a specialised protein complex (PfRCR) is secreted which forms a bridge for the parasite to facilitate entry into the next RBC.
Vaccine potential
By understanding this process, it is possible to design vaccines that can specifically target the protein complex to interrupt the progression of the disease. To fully comprehend the process, and how a vaccine can effectively target PfRCR to prevent the spread of the parasite into healthy cells, the study investigated the specific molecular function and structure of the protein complex.
Working with researchers from the University of Oxford’s Higgins Laboratory, RVC researchers, led by Dr Ellen Knuepfer, used ‘structure-guided’ molecular approaches to examine how PfRCR is assembled and functions as a whole within Plasmodium parasites during the invasion of RBCs.
To date, the project has made huge breakthroughs in the development of effective vaccine targets to ward against the contraction and progression of malaria. This includes rationalising regions within PfRCR to function as therapeutic targets in helping to reduce rates of death from the disease. However, work remains to be done to fully protect children against malaria and help eradicate the disease which currently remains one of the top 10 deadliest diseases in the world.
Fundamentals of invasion
Dr Ellen Knuepfer, lecturer in Vaccinology and RVC lead on this study, said: “Our study provided an exciting opportunity to discover the fundamentals of how malaria parasites invade human red blood cells – a key step in the parasite’s life cycle that can be therapeutically targeted.
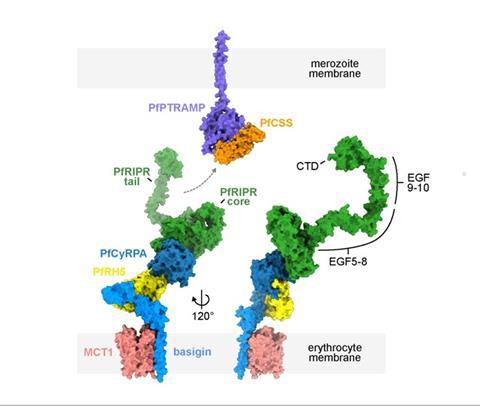
”Our results demonstrate that the essential multi-protein PfRCR complex does not function to create a molecular pore in the red blood cell membrane but rather as a molecular bridge, connecting the parasite to the red blood cell membrane during invasion. Excitingly, new structural insights will help us to rationally design therapeutics that target specific domains of this protein complex and hinder its function.”
Professor Matthew Higgins, EP Abraham Chair of Structural Biology at University of Oxford and head of Higgins Laboratory team, said: “We are delighted to be able to see, for the first time, this piece of molecular machinery which is essential for the malaria parasite to get inside our red blood cells. It was a pleasure to work with the RVC to test what the PfRCR does and to rule out some possible hypotheses for its essential role. Now that we know how PfRCR forms a bridge between parasite and erythrocyte, this will help us to design better vaccines against malaria.”







No comments yet