Tuberculosis (TB) disproportionately affects vulnerable populations including those with limited economic resources, HIV patients, those whose diet is deficient in nutrients and others.
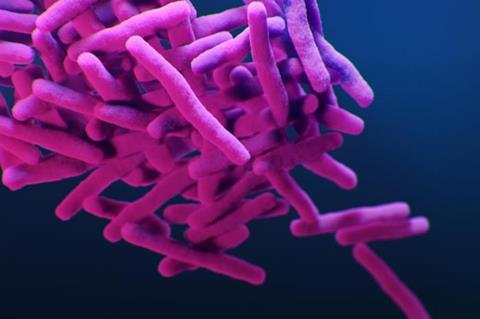
Resistant TB (MDR TB) does not respond to first line medications and is difficult to treat, requiring long regimens of 15-20 months that are associated with significant side effects and poor outcomes.
Recently, new six-month regimens have been shown to have better results than the long-term treatments, with improved quality of life and health equity. But these novel regimens have not yet been adopted widely in the United States.
Six-month regimen
In a study “Initial experience with BPaL based regimens for multidrug resistant tuberculosis treatment in Massachusetts” published in the International Journal of Tuberculosis and Lung Disease, researchers from Boston University Chobanian & Avedisian School of Medicine document that four patients at Boston Medical Center (BMC) Tuberculosis Clinic were cured of MDR TB with a six-month regimen that included bedaquiline, pretomanid and linezolid (BPaL).
“Our study showed that MDR TB can be successfully treated with a six-month regimen that includes only pills. The shorter duration of therapy and the lesser pill burden will result in a better quality of life, improving health equity and access to therapy for MDR TB patients,” said corresponding author Carlos Acuna-Villaorduna, MD, an adjunct assistant professor of infectious diseases. “BMC is the first site in Massachusetts to use this novel treatment for patients with resistant TB.”
“The novel BPaL regimen is a major advance as it achieves superior outcomes with less side effects in a significantly shorter period of time,” said Acuna-Villaorduna, who cited the collaboration between TB clinicians, public health nurses, pharmacists, microbiologists and public health leaders that allowed BMC to safely implement this cutting-edge treatment.
This study appeared online in The International Journal of Tuberculosis and Lung Disease.





No comments yet