Sanofi, DNDi and the HAT-r-ACC consortium announced the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) has adopted a positive scientific opinion of Fexinidazole Winthrop as first oral treatment of acute form of sleeping sickness (rhodesiense).
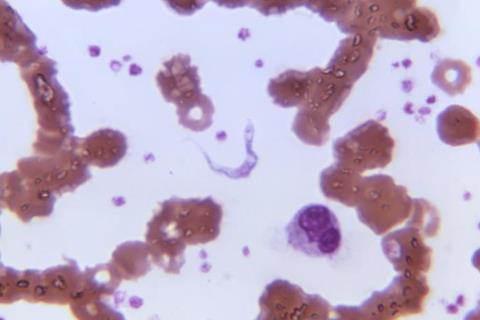
This positive opinion is for the treatment in adults and children six years of age or older and weighing at least 20 kg, of both first-stage (haemo-lymphatic) and second-stage (meningo-encephalitic) Trypanosoma brucei (T.b.) rhodesiense sleeping sickness, an acute and lethal form of this parasitic disease found in Eastern and Southern Africa.
This CHMP opinion follows an application by Sanofi under Article 58 and clinical trials in Malawi and Uganda led by the non-profit medical research organization Drugs for Neglected Diseases initiative (DNDi). In 2018, the CHMP first adopted a positive opinion of Fexinidazole Winthrop as the first all-oral treatment, in adults and children six years of age or older and weighing at least 20 kg, of both first-stage (haemo-lymphatic) and second-stage (meningo-encephalitic) of the more common T.b. gambiense form of sleeping sickness found in West and Central Africa.
Fatal without treatment
Sleeping sickness, or human African trypanosomiasis (HAT), is usually fatal without treatment. Both forms of sleeping sickness are transmitted by the bite of infected tsetse flies, which are found in 36 African countries. It causes neuropsychiatric symptoms, including aggressiveness, psychosis, a debilitating disruption of sleep patterns that have given this neglected disease its name, and ultimately, death.
‘The CHMP’s positive opinion is another step forward in Sanofi’s commitment to help deliver innovative treatments to vulnerable patient communities impacted by sleeping sickness, a deadly neglected tropical disease. By working with WHO and DNDi, we have made tremendous progress in improving treatment outcomes and simplifying treatment delivery. This partnership and our donation of Fexinidazole Winthrop through Foundation S, reflect our mission to provide innovative treatments to patients, no matter where they live,’ said Dietmar Berger, MD, PhD, Head of Development and Chief Medical Officer at Sanofi.
10 day treatment
For the T.b. rhodesiense variant, Fexinidazole Winthrop is indicated as a 10-day, once-daily oral treatment. Data from DNDi’s Phase 2/3 clinical trial were recently presented at the European Congress of Tropical Medicine and International Health and showed that Fexinidazole Winthrop was highly effective in treating the T.b. rhodesiense form of sleeping sickness and is a safe alternative to the existing drugs.
In periodic follow-up evaluations that continued for 12 months after treatment, only one patient (2.94%) with the advanced form of the disease had relapsed and required treatment with the arsenic derivative that is the standard of care for patients with the most severe stage of the disease.
‘T.b. rhodesiense sleeping sickness is a terrifying disease that progresses more rapidly than T.b. gambiense, killing quickly if untreated. Until now, due to the lack of innovation for this strain of sleeping sickness, old and toxic treatment options have to be administered in a hospital under strict surveillance. Having a simple and safer oral pill to treat this frightening disease will allow doctors to rapidly save lives. It will also help patients to trust the new treatment,’ said Dr Westain Nyirenda, Principal Investigator and physician at Rumphi Hospital in Malawi.
Zoonotic disease
While humans are the main host of T.b. gambiense, T.b. rhodesiense is a zoonotic disease, meaning that the infection can spread from animals to humans. Cattle and wild animals such as bushbucks and zebras are the most common reservoirs for this disease. Movements of these animals – potentially sparked by droughts or changes in climate – could put new populations at risk of T.b. rhodesiense sleeping sickness. In some cases, tourists visiting game reserves have been infected with T.b. rhodesiense.
‘We are already seeing how an all-oral treatment for T.b. gambiense sleeping sickness has simplified the treatment of this variant in countries like the Democratic Republic of Congo. Although there are comparatively few cases of T.b. rhodesiense, last year Ethiopia recorded its first cases since the 1970s. The drought at the time of the infections brought humans and cattle in closer proximity to tsetse flies’ habitat. Environmental changes could be one of the reasons behind this resurgence,’ said Dr Olaf Valverde Mordt, Clinical Project Leader for sleeping sickness at DNDi.
Update guidelines
The CHMP opinion paves the way for the update of WHO guidelines on treatment for sleeping sickness, and distribution by WHO of Fexinidazole Winthrop in African countries where T.b. rhodesiense is prevalent. Fexinidazole Winthrop will be donated to WHO by Foundation S, Sanofi’s philanthropic organization.
‘The impact of climate change extends to the shifting geographical spread of vector-borne diseases such as sleeping sickness, heightening the likelihood of spillover events where diseases transfer from animals to humans. These shifts disproportionately affect the most vulnerable communities, underscoring the urgency of sustained investment in programs addressing NTDs. This includes the development of innovative tools and improved treatment methods. We extend our gratitude to our partners DNDi for their research for the most neglected and Sanofi for their ongoing support and contribution to these vital efforts,’ said Dr Ibrahima Socé Fall, Director of Neglected Tropical Diseases at WHO.
Fexinidazole Winthrop has already been registered in the Democratic Republic of the Congo and Uganda as a treatment for T.b. gambiense and is recommended for use in a further 10 African countries: (Angola, Burkina Faso, Central African Republic, Chad, Congo, Côte d’Ivoire, Equatorial Guinea, Gabon, Guinea, and South Sudan).
Neglected disease
‘We congratulate the HAT-r-ACC consortium on developing a better treatment option for this truly neglected disease and are thrilled with the positive opinion by CHMP,’ said Dr Michelle Helinski, Senior Project Officer for Neglected Infectious Diseases at the European and Developing Countries Clinical Trials Partnership Association (EDCTP).
The DNDi clinical trial for T.b. rhodesiense was conducted by the HAT-r-ACC Consortium, with funding from the European and Developing Countries Clinical Trials Partnership Association (EDCTP2) programme supported by the European Union; Fundação para a Ciência e a Tecnologia from Portugal; Swiss Agency for Development and Cooperation (SDC), from Switzerland; Médecins Sans Frontières International; and UK International Development, United Kingdom; and other private foundations and individuals.
Topics
- Clinical & Diagnostics
- Committee for Medicinal Products for Human Use
- Dietmar Berger
- Disease Treatment & Prevention
- DNDi
- European Medicines Agency
- Fexinidazole Winthrop
- HAT-r-ACC
- human African trypanosomiasis
- Ibrahima Socé Fall
- Medical Microbiology
- Michelle Helinski
- Middle East & Africa
- Olaf Valverde Mordt
- One Health
- Parasites
- People News
- Rumphi Hospital
- Sanofi
- sleeping sickness
- Trypanosoma brucei (T.b.) rhodesiense
- UK & Rest of Europe
- Veterinary Medicine & Zoonoses
- Westain Nyirenda






No comments yet