Sebastian Schornack’s research team is working to identify and characterise new arbuscular mycorrhiza (AM) effectors – proteins secreted by symbiotic AM fungi to facilitate colonisation of plant roots. As part of this they are also investigating whether pathogenic and symbiotic fungi are using similar or different strategies.
Using the AlphaFold2 AI computer programme that predicts protein structures, the team is the first to compare secreted proteins from symbiotic and pathogenic fungi. They found that symbiotic fungi encode the same family of proteins that pathogenic fungi use to interact with and colonise plants. The findings were published in New Phytologist.
Huge diversity
“There is a huge diversity in the genetic code of effector proteins in closely related microbes, so it is even more difficult to compare protein sequences between pathogens and symbionts that are more distantly related,” said Dr Albin Teulet, who is first author of the research.
“Instead, we took advantage of the recent development of AlphaFold2 to do a comparison between the secreted protein structures from a symbiotic fungus and pathogenic fungus. This led to the discovery that there is a group of structurally very similar proteins previously known from pathogens that are also present – often in greater numbers – encoded in the genomes of symbiotic fungi.”
Plant-fungi relationships have a dramatic effect on plant health in natural ecosystems and agriculture – both positive and negative. AM fungi are the most common symbiotic association of plants with microbes, colonising plant roots to promote nutrient uptake, enhance plant growth and even help fend off pathogens, while pathogenic fungi, such as strains of Fusarium oxysporum, are some of the world’s most devastating pathogens with a wide host range and are currently threatening to wipe out Cavendish banana and oil palm plantations. Fusarium also has lent its name to the group of proteins the team have found: Fusarium oxysporum lycopersici dual-domain proteins (FOLDs).
Family of effectors
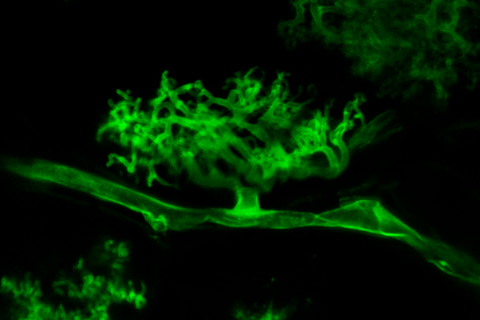
The researchers classified all the proteins secreted by the symbiotic AM fungus Rhizophagus irregularis and identified a large family of FOLD-like effectors (MycFOLDs) with high structural similarity to the FOLD proteins produced by the Fusarium oxysporum pathogen.
FOLD effectors are only found in the genomes of fungi that form associations with living plants. This suggests that these proteins underpin universal mechanisms enabling both pathogens and symbiotic fungi to live within plants.
Dr Schornack said discovering that plant symbiotic and pathogenic fungi were using some of the same tools provided a new perspective on our understanding of fungal symbionts: “The apparently exclusive presence of FOLD/MycFOLDs across unrelated plant-colonising fungi supports the hypothesis that FOLD proteins act as effectors during plant colonisation of both symbiotic and pathogenic fungi.”
“If symbiotic and pathogenic fungi are using the same tools, this means devising ways to enhance AM relationships in crops might make the crops more susceptible to pathogens at the same time. However, these findings now open new questions for us to explore, such as why do symbiotic fungi have these proteins that pathogens also have and what do they actually do?”





No comments yet