More than a decade after the Nagoya Protocol, which aims to fairly share the benefits of utilizing genetic resources, became law, microbiologists and other scientists still face practical challenges and confusion.
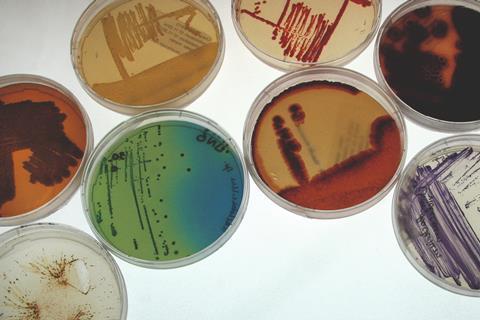
A new guide published by a team of European microbiologists provides universally applicable frameworks for anyone working with biological resources.
The guidance is outlined in ‘How to do the Nagoya Protocol: common misconceptions, challenges and best practices for access and benefit-sharing compliance’ which appears in Sustainable Microbiology, an Applied Microbiology International publication.
“Science has changed a lot in the past decade and terms like ‘(don’t do) helicopter science’ and ‘equity in science’ are much more common. Yet many microbiologists don’t actually know that this is much more than just best practice. There is binding international law that regulates how international research collaborations should be done,” the authors say.
“The Nagoya Protocol is the legal basis and we all need to comply with it. But it is hard to understand at first and, in practice, sometimes quite challenging to be in full compliance. So researchers need guidance on what to do and how to overcome known issues.”
International team
The study included a diverse international team of authors from culture collections, research institutes, universities, and research consortia. The collaboration, led by Leibniz Institute DSMZ, brought together several large Horizon Europe consortia: Microbiome Biobanking Enabler (MICROBE), the German Nagoya Protocol HuB and contributors from Microbes-4-Climate, European Viral Outbreak Response Alliance, and other international groups including MIRRI-ERIC, the European Culture Collections Organisation, Global Microbiome Conservancy, Afribiota, and the Centre for Agriculture and Bioscience International.

The team have developed a ‘policy in practice’ framework for how “to do” the Nagoya Protocol as it applies to microbiological (and broader biological) research - including best practices, a five-step compliance workflow, practical checklists, and real-world case studies. This work is complemented by a shorter policy briefing on broader regulatory and international legal issues affecting microbiologists in the same journal issue.
Illegal research
“If you don’t know about the Nagoya Protocol and follow the resulting national laws, it is quite likely you will be conducting illegal research in some of your international collaborations,” warn the authors.
“Don’t do this! Learn more and figure out how to be legally compliant! It can really damage your reputation and that of your institution and even result in legal fines and other consequences.”
For example, if a researcher has biological samples, what are the rules that govern accessing them? Who do they need to ask for permission, how does that work, and where can they get help?
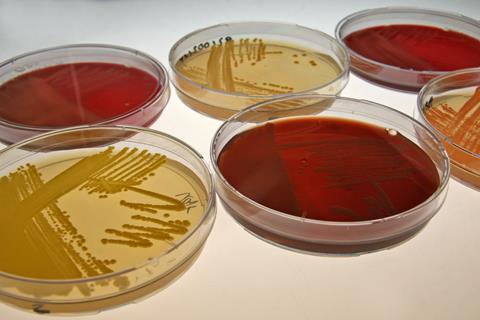
These legal rules - known as access and benefit-sharing or ABS for short - affect daily lab workflows, culture collections, and biobank management. Uncertainty causes delays, legal risk, prevented sample sharing, blocked strain deposition, and impeded taxonomic publications.
Common misunderstandings
The team collated and analyzed common misunderstandings and operational ABS challenges from contributor experience and wider networks; streamlined key compliance steps into a simple five-step workflow; and produced practical checklists and illustrated the approach with three representative case studies from international projects and legal practice.

“ABS compliance is achievable and scalable when anticipated early and integrated into project design,” they reassure researchers.
“Critical success factors include clear documentation, flexible permit conditions, explicit consent for future uses/sharing, and sustained engagement with provider-country authorities and, perhaps most importantly, in-country collaboration partners.
“However, persistent barriers still exist more than ten years after entry into force: inconsistent national procedures, language differences, unclear legal definitions for microorganisms, and limited ABS training among researchers. Researchers should leverage professional networks, local partners, and institutional/online ABS support hubs.”
Legal uncertainty
One finding was that ABS restrictions are increasingly affecting core scientific practices, with legal uncertainty over human-associated microbes sometimes forcing permit renegotiation or renewed participant consent long after collection.
However, researchers are generally willing to engage constructively with ABS regimes and negotiate fair, transparent agreements.

The authors said poor ABS planning can directly compromise scientific output, but robust ABS workflows protect researchers, institutions, collaborations, and reputations.
“Clear compliance processes facilitate material sharing, reproducibility, and equitable partnerships. At the end, Nagoya compliance supports trust, sustainability, responsible global research and support biodiversity conservation.”
Provider countries
They recommend that provider countries (those giving access to biodiversity samples) should promote standardization (model agreements, benefit-sharing clauses, due-diligence templates).
ABS/Nagoya training should be integrated into the life science curricula and culture/biobank management, and ongoing dialogue should be maintained between scientists and national authorities, especially for (but not limited to) human-associated microbes.
“Make ABS compliance resources simple, digital, and multilingual to reduce administrative burdens for international teams,” they suggest.
The lead authors are Davide Faggionato and Melania Muñoz-García; corresponding author is Amber H. Scholz.
The project is supported / funded by: European Commission (Horizon Europe and NextGenerationEU); German Alliance of Scientific Organizations; German Science Foundation; Massachusetts Institute of Technology; European Research Council; Swiss National Science Foundation; co-funded by the European Union.
‘How to do the Nagoya Protocol: common misconceptions, challenges and best practices for access and benefit-sharing compliance’ is published in Sustainable Microbiology.
Topics
- access and benefit-sharing
- Afribiota
- Amber H. Scholz
- Applied Microbiology International
- Centre for Agriculture and Bioscience International
- Community
- Davide Faggionato
- Diversity & Inclusion
- Economic Equality
- European Culture Collections Organisation
- European Viral Outbreak Response Alliance
- genetic resources
- German Nagoya Protocol HuB
- Global Microbiome Conservancy
- Leibniz Institute DSMZ
- Melania Muñoz-García
- Microbes and Culture
- Microbes-4-Climate
- Microbiome Biobanking Enabler
- MIRRI-ERIC
- Nagoya Protocol
- Policy
- Research News
- UK & Rest of Europe





No comments yet