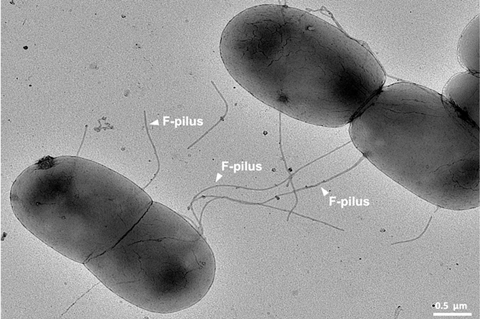
Gut bacteria form extracellular appendages called F-pili to connect to each other and transfer packets of DNA, called genes, that allow them to resist antibiotics. It was thought that the harsh conditions inside human and animal guts, including turbulence, heat, and acids, would break the F-pili, making transfer more difficult.
However, new research by a team led by Imperial College London researchers has shown that the F-pili are actually stronger in these conditions, helping the bacteria transfer resistance genes more efficiently, and to clump into ‘biofilms’ – protective bacterial consortia – that help them fend off antibiotics.
The discovery shows why it can be so difficult to tackle drug-resistant bacteria, but does provide a possible avenue for tackling the problem. The super-polymer structures the bacteria use to transfer genes could also be exploited for precise drug delivery in future medicine. The results are published in Nature Communications.
First author Jonasz Patkowski, from the Department of Life Sciences at Imperial, said: “The death toll from antimicrobial resistance is expected to match cancer by 2050, meaning we urgently need new strategies to combat this trend. Much of the spread of resistance is driven by bacteria swapping genes, so detailed understanding of this process could lead to new ways to interrupt it.”
Not so fragile
Different classes of bacteria use different types of pili to transfer genes in a process called conjugation. A classic experiment seemed to show that this process was fragile and could be interrupted by agitation, but this left a mystery: why do so many bacteria living in harsh conditions like guts use these systems if they are so fragile?
The team therefore set out to test this assumption. By shaking E. coli bacteria while they used F-pili during conjugation, they discovered that agitation actually increased the efficiency of gene transfer between bacteria. They also observed that after transferring genes, the conjugated bacteria in shaken conditions clumped together more easily to form biofilms, which protect inner bacteria from the surrounding antibiotic molecules.
To determine how the F-pili are able to do this, the team subjected them to a strength test by mounting a bacterium on a stage, connecting a glass bead using ‘molecular tweezers’ to the end of one of its F-pili, and pulling. The F-pili proved highly elastic, with spring-like properties that prevented them from breaking.
They also tested the F-pili’s ability to withstand other common gut conditions, subjecting them to sodium hydroxide, urea, and excessively high temperatures of 100°C – all of which the F-pili survived.
Molecular properties
The team then went a step further, looking at the F-pili on a molecular level to see what gives them these incredible properties. They are primarily made up of F-pilin ‘subunits’ with interlinked phospholipid molecules.
By modelling the F-pili without the phospholipids, the team showed how important these molecules are for the structure’s springiness and elastic strength. Repeating the pulling experiment revealed that the subunits quickly disassemble without the phospholipids supporting them, proving their novel role as a ‘molecular glue’ in long biopolymers.
Lead researcher Dr Tiago Costa, from the Department of Life Sciences at Imperial, said: “Making F-pili is very costly to the bacteria in terms of resources and energy, so it’s no surprise they are worth the effort. We have shown how F-pili accelerate the spread of antibiotic resistance and biofilm formation in turbulent environments, but the challenge now is to find ways to combat this very efficient process.”
While it would be advantageous to break F-pili in pathogenic bacteria, their properties could be helpful if we can engineer them for use in, for example, drug delivery. Patkowski explained: “It’s hard to find a tubular appendage with such strong properties. Bacteria use it to transfer genes, but if we could mimic these properties, we could use similar structures to precisely deliver drugs where they are needed in the body.”





No comments yet