Peruvoside, a plant-based compound that is commonly used to treat heart failure, has been discovered to be able to prevent up to 12 medically important viruses, all originating from different virus families.
These viruses are of medical significance as they cause prevalent viral diseases, such as SARS-CoV-2, Hand, Foot and Mouth Disease (HFMD) and influenza. When modified, peruvoside acts on GBF1, a protein that is crucial for the replication and production of virus in the body, by disabling its functionality so that production of more virus is stopped.
Led by Associate Professor Justin Chu from the Infectious Diseases Translational Research Programme and Department of Microbiology and Immunology at the Yong Loo Lin School of Medicine, National University of Singapore (NUS Medicine), the study aimed to find a therapeutic that is effective in killing a wide range of viruses.
Screening compounds
Thousands of compounds were screened using a high-throughput screening test, normally used in drug discovery processes. Peruvoside was identified as the ideal candidate and was found to possess broad-spectrum antiviral properties to prevent the replication of a number of infectious viruses, with minimal side effects.
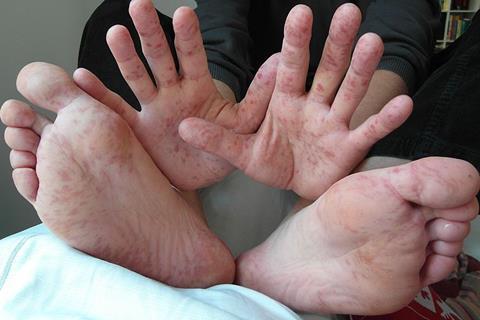
In their study, the team evaluated the efficacy of peruvoside in fighting enterovirus, the virus that causes HFMD. At a treatment dosage level of 0.5 milligrams, the results showed 100% efficacy in protection with no detectable traces of virus in tissues, the region usually affected by the enterovirus.
The team also measured the toxicity of the peruvoside drug using the LDH assay, which is revealed based on the level of liver enzymes in the blood stream. An inflamed liver or damage to liver cells would lead to elevated levels of liver enzymes in the blood test. In the study, a very low level of toxicity was detected following the administration of peruvoside at different concentrations.
Targeting intruders
In the field of infectious diseases, there are typically two ways to target the intruding virus. The first method is targeting the virus particles using antibodies, at the start of the infection process.
The second method is targeting the infected cells where the virus replicates itself, which is known as the viral factories. The virus begins its infectious stage at the entry point of the cell. Once the virus gets to the entry point, it releases its genetic material into the cells to form viral factories, that replicate and produce more copies of the virus, developing into an infectious disease within the body.
Many commonly-known viruses of medical importance such as SARS-CoV-2, Dengue virus, Zika virus, Chikungunya virus and Enterovirus are known as positive-sense RNA viruses. For this group of viruses, the virus replication cycle takes place exclusively in the cytoplasm of the cell, instead of the cell nucleus.
The virus decodes and releases its genome to be replicated, building up viral factories within the cytoplasm to ensure virulence.
Virus replication
Surprisingly, the team found that peruvoside also has the ability to prevent virus replication of other viruses that cause Herpes Cold Sores, H1N1 Human Influenza A and the common flu, with minimal side effects. These viruses are not in the same positive-sense RNA virus group that the team initially set out to test.
Associate Professor Justin Chu is confident of the benefits of peruvoside as the next effective drug to fight against existing and emerging viruses.
Speaking as Principal Investigator of the study, Assoc Prof Chu said: “This study shows great significance for peruvoside to be considered as a general cellular target and an anti-viral therapeutic for medically important virus-mediated diseases, especially when no vaccine and therapeutic is available.
“For the next stage of this study, the team aims to modify and improve the pharmacological and safety profile of peruvoside so that it can be eligible for clinical trials and safe for consumption.”
This study is published in Acta Pharmaceutica Sinica B.







No comments yet