Researchers have developed an automated version of the viral plaque assay, the gold-standard method for detecting and quantifying viruses. The new method uses time-lapse holographic imaging and deep learning to greatly reduce detection time and eliminate staining and manual counting.
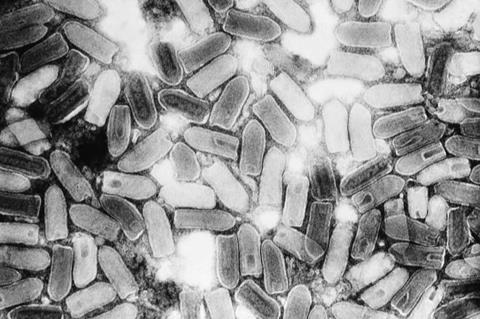
This advance could help streamline the development of new vaccines and antiviral drugs. Yuzhu Li from Ozcan Lab at the University of California, Los Angeles (UCLA), will present the research at Frontiers in Optics + Laser Science (FiO LS), which will be held 9 – 12 October 2023 at the Greater Tacoma Convention Center in Tacoma (Greater Seattle Area), Washington.
“By cutting down the detection time compared to traditional viral plaque assays, this technique might help expedite vaccine and drug development research by significantly reducing the detection time needed and eliminating chemical staining and manual counting entirely, explains Li. “In the event of a new virus outbreak, vaccines or antiviral treatments could be developed, tested, and made available to the public at a significantly accelerated rate, resulting in a faster response time to virus-induced health emergencies.”
Time consuming
Although the viral plaque assay is a cost-effective way to assess virus infectivity and quantify the amount of a virus in a sample, it is time consuming to perform. Samples are first diluted and then added to cultured cells.
If the virus kills the infected cells, a region free of cells — a plaque — develops. Experts then manually count the stained plaque-forming units (PFUs), a process that is susceptible to staining irregularities and human counting errors.
The new stain-free automated viral plaque assay system replaces manual plaque counting with a lens-free holographic imaging system that images the spatiotemporal features of PFUs during incubation. A deep learning algorithm is then used to detect, classify and locate PFUs based on changes observed.
Faster approach
To show the efficacy of their system, the researchers infected cultured cells with the Vesicular stomatitis virus. After just 20 hours of incubation, the automated system detected more than 90% of the viral PFUs without any false positives. This was much faster than the traditional plaque assay, which requires 48 hours of incubation for this virus.
They also applied the automated approach to herpes simplex virus type-1 and encephalomyocarditis virus. They demonstrated even shorter incubation times for these viruses, saving an average of around 48 and 20 hours, respectively.
No false positives
The researchers report that no false positives were detected across all time points. In addition, because the system can identify individual PFUs during their early growth, before the formation of PFU clusters, it can be used to analyze viral samples with about 10 times higher virus concentrations than traditional approaches.
“As for the next steps, UCLA researchers are improving their system design to further increase its sensitivity and specificity for various types of viruses, paving the way for broad adoption in laboratory and industrial settings, said Li. “They are also exploring other potential applications of this technique in virology research for high-throughput and cost-effective screening of antiviral drugs.”
Topics
- Artificial Intelligence & Machine Learning
- Clinical & Diagnostics
- encephalomyocarditis virus
- Future Technologies
- herpes simplex virus type-1
- Innovation News
- Medical Microbiology
- One Health
- Ozcan Lab
- plaque-forming units
- time-lapse holographic imaging
- University of California
- USA & Canada
- Vesicular stomatitis
- viral plaque assay
- Viruses
- Yuzhu Li





No comments yet