Getting results from a blood test can take anywhere from one day to a week, depending on what a test is targeting. The same goes for tests of water pollution and food contamination, and in most cases, the wait time has to do with time-consuming steps in sample processing and analysis.
Now, MIT engineers have identified a new optical signature in a widely used class of magnetic beads, which could be used to quickly detect contaminants in a variety of diagnostic tests. For example, the team showed the signature could be used to detect signs of the food contaminant Salmonella.
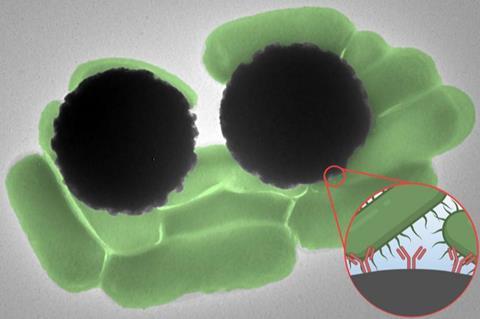
The so-called Dynabeads are microscopic magnetic beads that can be coated with antibodies that bind to target molecules, such as a specific pathogen. Dynabeads are typically used in experiments in which they are mixed into solutions to capture molecules of interest. But from there, scientists have to take additional, time-consuming steps to confirm that the molecules are indeed present and bound to the beads.
Raman signature
The MIT team found a faster way to confirm the presence of Dynabead-bound pathogens, using optics, specifically, Raman spectroscopy. This optical technique identifies specific molecules based on their “Raman signature,” or the unique way in which a molecule scatters light.
The researchers found that Dynabeads have an unusually strong Raman signature that can be easily detected, much like a fluorescent tag. This signature, they found, can act as a ‘reporter’. If detected, the signal can serve as a quick confirmation, within less than an hour, that a target pathogen is indeed present in a given sample.
The team is currently working to develop a portable device for quickly detecting a range of bacterial pathogens, and has reported their results in a special issue of the Journal of Raman Spectroscopy.
Narrowing the suspects
“This technique would be useful in a situation where a doctor is trying to narrow down the source of an infection in order to better inform antibiotic prescription, as well as for the detection of known pathogens in food and water,” says study co-author Marissa McDonald, a graduate student in the Harvard-MIT Program in Health Sciences and Technology.
“Additionally, we hope this approach will eventually lead to expanded access to advanced diagnostics in resource-limited environments.”
Study co-authors at MIT include Postdoctoral Associate Jongwan Lee; Visiting Scholar Nikiwe Mhlanga; Research Scientist Jeon Woong Kang; Tata Professor Rohit Karnik, who is also the associate director of the Abdul Latif Jameel Water and Food Systems Lab; and Assistant Professor Loza Tadesse of the Department of Mechanical Engineering.
Oil and water
Looking for diseased cells and pathogens in fluid samples is an exercise in patience.
“It’s kind of a needle-in-a-haystack problem,” Tadesse says.
The numbers present are so small that they must be grown in controlled environments to sufficient numbers, and their cultures stained, then studied under a microscope. The entire process can take several days to a week to yield a confident positive or negative result.
Both Karnik and Tadesse’s labs have independently been developing techniques to speed up various parts of the pathogen testing process and make the process portable, using Dynabeads.
Microscopic beads
Dynabeads are commercially available microscopic beads made from a magnetic iron core and a polymer shell that can be coated with antibodies. The surface antibodies act as hooks to bind specific target molecules. When mixed with a fluid, such as a vial of blood or water, any molecules present will attach onto the Dynabeads.
Using a magnet, scientists can gently coax the beads to the bottom of a vial and filter them out of a solution. Karnik’s lab is investigating ways to then further separate the beads into those that are bound to a target molecule, and those that are not. “Still, the challenge is, how do we know that we have what we’re looking for?” Tadesse says.
Invisible to the eye
The beads themselves are not visible by eye. That’s where Tadesse’s work comes in. Her lab uses Raman spectroscopy as a way to “fingerprint” pathogens. She has found that different cell types scatter light in unique ways that can be used as a signature to identify them.
In the team’s new work, she and her colleagues found that Dynabeads also have a unique and strong Raman signature that can act as a surprisingly clear beacon.
“We were initially seeking to identify the signatures of bacteria, but the signature of the Dynabeads was actually very strong,” Tadesse says. “We realized this signal could be a means of reporting to you whether you have that bacteria or not.”
Testing beacon
As a practical demonstration, the researchers mixed Dynabeads into vials of water contaminated with Salmonella. They then magnetically isolated these beads onto microscope slides and measured the way light scattered through the fluid when exposed to laser light. Within half a second, they quickly detected the Dynabeads’ Raman signature — a confirmation that bound Dynabeads, and by inference, Salmonella, were present in the fluid.
“This is something that can be used to rapidly give a positive or negative answer: Is there a contaminant or not?” Tadesse says. “Because even a handful of pathogens can cause clinical symptoms.”
The team’s new technique is significantly faster than conventional methods and uses elements that could be adapted into smaller, more portable forms — a goal that the researchers are currently working toward. The approach is also highly versatile.
Proof of concept
“Salmonella is the proof of concept,” Tadesse says. “You could purchase Dynabeads with E.coli antibodies, and the same thing would happen: It would bind to the bacteria, and we’d be able to detect the Dynabead signature because the signal is super strong.”
The team is particularly keen to apply the test to conditions such as sepsis, where time is of the essence, and where pathogens that trigger the condition are not rapidly detected using conventional lab tests.
“There are a lot cases, like in sepsis, where pathogenic cells cannot always be grown on a plate,” says Lee, a member of Karnik’s lab. “In that case, our technique could rapidly detect these pathogens.”
This research was supported, in part, by the MIT Laser Biomedical Research Center, the National Cancer Institute, and the Abdul Latif Jameel Water and Food Systems Lab at MIT.







No comments yet