Cells rely on biomolecular condensates to coordinate essential biological processes without surrounding membranes. These droplet-like dynamic assemblies control the way in which DNA is turned into proteins, help clear cellular waste to prevent toxicity and disease, and can even suppress cancerous tumors. Because they behave like liquids, able to merge, flow and rapidly exchange their contents, scientists long assumed condensates lacked internal organization and functioned as a simple liquid.
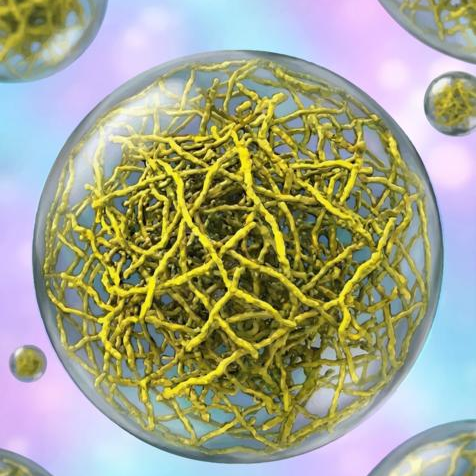
A study published in Nature Structural and Molecular Biology on February 2, 2026, overturns this assumption. Scientists at Scripps Research discovered that some condensates are built from intricate networks of thread-like protein filaments. Far from being shapeless droplets, these structures contain a defined internal architecture that is essential for cellular function. The findings suggest new possibilities for therapeutic intervention in diseases such as cancer and neurodegeneration.
“Ever since we realized that disruptions in condensate formation are at the heart of many diseases, it has been challenging to target them therapeutically because they appeared to lack structure—there were no specific features for a drug to latch onto,” says Keren Lasker, associate professor at Scripps Research and senior author of the study. “This work changes that. We can now see that some condensates have an internal architecture, and that, importantly, this structure is required for function, opening the door to targeting these membrane-less assemblies much like we target individual proteins.”
Bacterial protein
To understand how condensates achieve compartment-like behavior without membranes, the Lasker lab focused on a bacterial protein called PopZ. In certain rod-shaped bacteria, this protein forms condensates at the cell poles (the rounded ends of the cell), where they organize proteins for cell division.
In close collaboration with Scripps Research professor Ashok Deniz and assistant professor Raphael Park, co-corresponding authors on this study, the researchers used cryo-electron tomography (cryo-ET), essentially a CT scan for molecular structures, to visualize PopZ condensates at unprecedented resolution. They found that the filaments assemble through a precise, stepwise process, forming structural scaffolds that give the condensate its physical properties.
READ MORE: Scientists reveal how first cells could have formed on Earth
READ MORE: The very first structural images of a tuberculosis-fighting virus
The structural discovery was only the beginning. Using single-molecule Förster resonance energy transfer (FRET), a technique that measures tiny changes in distance within individual proteins by tracking energy transfer between fluorescent tags, the team revealed that PopZ molecules adopt distinct conformations depending on whether they were inside or outside a condensate.
“Realizing that protein conformation depends on location gives us multiple ways to engineer cellular function,” says Daniel Scholl, first author and former postdoctoral researcher in the Lasker and Deniz labs.
Filament formation
To determine whether filaments were structurally incidental or biologically essential, the team introduced a mutant that abolished filament formation. The resulting condensates were markedly more fluid, with reduced surface tension. When tested in living bacteria, growth ceased entirely, and DNA segregation failed—demonstrating that the physical properties of the condensate, not simply its molecular components, are required for cellular function.
The broader implications reach well beyond bacterial biology. In human cells, similar filament-based condensates help perform two critical functions: removing damaged or toxic proteins and preventing uncontrolled cell growth. When the cleanup condensates fail, harmful proteins can accumulate—a hallmark of neurodegenerative diseases such as ALS. When the growth-regulating condensates malfunction, the safeguards against tumor formation break down, contributing to cancers including prostate, breast and endometrial.
“By demonstrating that condensate architecture is both definable and functionally critical, the work raises the possibility of designing therapies that act directly on condensate structure and correct the underlying disorganization that allows disease to take hold,” says Lasker.






No comments yet