In eastern Africa, malaria parasites have developed resistance to artemisinins, the backbone of current treatment regimens, a development that could dramatically worsen malaria’s impact if partner drugs fail in the future.
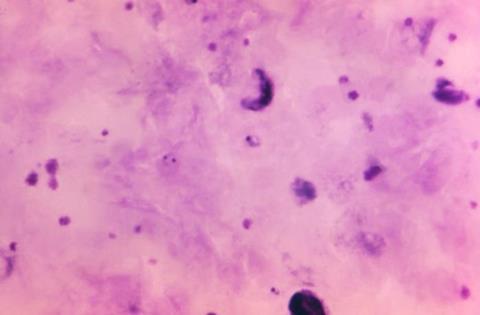
The finding from studies in Eritrea was reported in the New England Journal of Medicine by a team of researchers led by Didier Ménard, PhD, of the Université de Strasbourg/Institut Pasteur in France and including Columbia University microbiologist David Fidock, PhD, the C.S. Hamish Young Professor of Microbiology & Immunology and professor of medical sciences in the Vagelos College of Physicians and Surgeons.
Treatment of malaria depends on artemisinin drugs paired with a partner antimalarial. These drug combinations have been highly effective treatments for non-severe cases since the early 2000s and usually clear the malaria parasites from a patient’s blood after three days of treatment.
Resistance to artemisinins
But Plasmodium falciparum parasites are developing drug resistance, which threatens to roll back the progress made against malaria between 2000 and 2015, when deaths from the disease in Africa dropped by 66%. Resistance to artemisinins first emerged in Southeast Asia in 2009, followed soon after by resistance to partner drugs.
By 2016, the treatment failure rate in some parts of Southeast Asia had reached 85%. Resistance to the artemisinin components is caused by mutations in the P. falciparum parasite gene Pfkelch13.
With drug-resistant malaria, what happens in Southeast Asia often occurs in Africa with a decade-long delay, either because resistant parasites cross over to Africa or the same mechanism of resistance takes longer to emerge and establish itself in high-transmission African settings. More than 95% of all deaths from malaria occur in Africa, and any increase in drug resistance there is alarming.
Drug failure
In the new study, Ménard’s group and colleagues from the Ministry of Health in Eritrea assessed the effectiveness of artemisinin-based combination therapies in nearly 1,000 patients in Eritrea between 2016 and 2019.
The researchers found that effectiveness of the drug therapy declined during that time: The drugs failed to clear parasites in 0.4% of patients in 2016, rising to 4.2% in 2019, crossing the WHO threshold for declaring resistance.
The researchers also found that by 2019, about one in five patients was infected with artemisinin-resistant Pfkelch13 mutant parasites.
The Columbia team led by Fidock then performed genetic experiments with parasites grown in a laboratory and showed that the most common Pfkelch13 mutation identified in Eritrea is directly responsible for the artemisinin resistance.
Genetic surveillance
The question now is how widespread the mutations in Pfkelch13 are across Africa. “We’re not looking at a new strain that’s recently taken over. It’s just taken this long to detect,” Fidock says. “Central and western Africa have high malaria burdens, but we don’t know what’s happening there and need more genetic surveillance and therapeutic efficacy studies.”
The situation in Eritrea is even more alarming, the study found, because many of the parasites harbor genetic deletions that make the parasite undetectable with the most common rapid diagnostic test for malaria.
About 17% of patients in Eritrea would test negative for the disease with this test, which is no longer used in Eritrea but is commonly used throughout Africa. The spread of these test-negative parasites would pose serious impediments to proper diagnosis.
Fatal outcomes
“That means if somebody goes to a clinic with symptoms, but the test comes back negative for malaria, they’re not going to be prescribed the right treatment,” Fidock says. “Their symptoms may get worse, or they may die. This risk is compounded by the fact that artemisinins are used alone to treat severe malaria, where drugs have to be delivered intravenously. Parasites with the mutant Pfkelch13 gene may not be quickly eliminated, increasing the risk of a fatal outcome. Clinicians across the region need to be aware that patients who test negative may indeed have malaria.”
“Unfortunately, our study has revealed that resistance has established a firm foothold in the Horn of Africa, which makes it more likely that the partner drugs will fail next because they are not being eliminated by the artemisinin, and malaria cases and deaths may start to spike,” says Ménard.
The situation is not yet catastrophic, because the parasites have not developed resistance to the partner drugs used in artemisinin therapy.
“But if those partner drugs fail, the situation could quickly worsen,” Fidock says. “There are enormous efforts underway to develop new drugs, but right now there are very limited options available.”
Topics
- Antimicrobial Resistance
- artemisinin
- Columbia University
- David Fidock
- Didier Ménard
- Eritrea
- Infectious Disease
- Institut Pasteur
- malaria
- Middle East & Africa
- One Health
- Parasites
- Pfkelch13
- Plasmodium falciparum
- Research News
- UK & Rest of Europe
- Université de Strasbourg
- USA & Canada
- Vagelos College of Physicians and Surgeons





No comments yet