The chemical synthesis of the powerful anticancer drug Vinblastine poses major challenges, owing to its complex three-dimensional structure. Vinblastine is a heterodimer of the chemical compounds catharanthine and vindoline, and currently available methods for the mass production of vinblastine are both costly and unsustainable.
A research team from Zhejiang University, China, led by Dr. Jiazhang Lian and Dr. Di Gao, have made a breakthrough by identifying a novel method for the biological synthesis of catharanthine and vindoline—the two pharmacologic precursors of vinblastine. The biosynthesis of these two precursors occurs via a 31-step-long reaction in Catharanthus roseus —a medicinal plant also known for its ornamental value.
Drawing inspiration from this study, the research team genetically engineered Saccharomyces cerevisiae, commonly known as yeast cells, and utilized them to produce vindoline and catharanthine.
The chemical/biological coupling of vindoline and catharanthine was then used to obtain vinblastine. These findings were published in BioDesign Research.
Dr. Lian, who serves as a Principal Investigator at Zhejiang University’s College of Chemical and Biological Engineering, says: “On the basis of a platform strain with a sufficient supply of precursors and cofactors for biosynthesis, we could successfully optimize the biosynthetic pathways to produce vindoline and catharanthine.
”Using shake flask fermentation, our engineered yeast strains were able to produce catharanthine and vindoline at a titer of 527.1 μg/L and 305.1 μg/L, respectively, without accumulating detectable amount of pathway intermediates.”
Genome modification
To modify the genome of the yeast cells, the team used the popular “CRISPR-Cas9” technique, further tweaking the technique to get rid of certain production bottlenecks. For instance, introducing an extra copy of the genes that coded for certain key enzymes significantly boosted the overall yield of catharanthine from 225.3 μg/L to 527.1 μg/L.
Similarly, when the team realized that the production of tabersonine - a reaction intermediate that ultimately gets converted to vindoline - was lower than that of catharanthine, they swiftly removed this bottleneck by introducing extra copies of genes coding for the associated enzymes. This increased the yield of tabersonine from 9.0 μg/L to 18.9 μg/L. A similar strategy was used to increase the yield of vindoline.
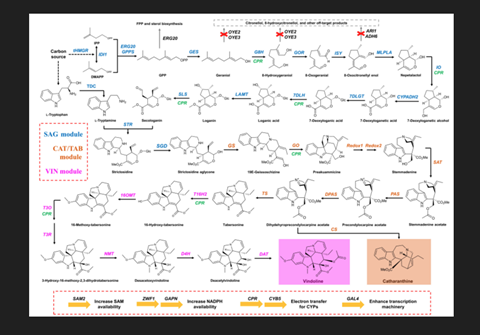
Discussing the future implications of these findings, Dr. Lian adds: “The production of vinblastine using microbial cell factories holds great promise for its large-scale and cost-effective production. In fact, it can act as a representative example for the production of additional valuable plant products in yeast.”







No comments yet