Space…the huge void! A hostile environment and a seemingly infinite expanse! People have always looked at space in awe, as another world that is beyond reach but was always intrigued to discover.
Since the first time man travelled to space in 1961, giant leaps have been made. It has now been visited by the brave few which we call astronauts. However we can explore space in a different way without putting on spacesuits, but rather our lab coats, even though wearing a spacesuit is rather cool. The International Space Station (ISS) is a huge laboratory that can be used and exploited to perform research in space, the results of which can help us understand life on Earth and apply them to improve our knowledge and way of life. Here we are going to explain our research…Project Maleth, Malta’s first biomedical experiment in space.
Diabetes and Diabetic Foot Ulcers
More than half a billion people are suffering from diabetes worldwide. Malta, a small island in the middle of the Mediterranean Sea has its fair share, with 11.2% of our population suffering from this disease. Diabetic patients are prone to ischaemia and peripheral neuropathy as well as an impaired immunity. These can lead to the formation of diabetic foot ulcers (DFUs) which tend to heal very slowly, becoming chronic and consequently infected. Normal clinical signs and symptoms associated with disease such as local tenderness, warmth and swelling are usually absent or reduced in these patients and therefore this contributes to a delay in them seeking medical attention. In fact, 44-68% of infected DFUs continue on to develop osteomyelitis, often requiring amputations affecting the patient’s quality of life as well as their loved ones. In Malta approximately 400 amputations, both minor and major, occur every year which for quite a small country is surprisingly high. Due to its chronicity, diabetes has a huge impact on the healthcare system and the numbers are only predicted to increase. Therefore we have decided to focus our research on this disease, most specifically on the microbiome that inhabits diabetic foot ulcers.

The skin microbiome and DFUs
The skin microbiome is a complex and diverse microbial community. Various studies have shown that the skin microbiome varies amongst individuals but not only that, it is also site specific. By means of molecular techniques we are now able to delve deeper and understand the composition of this microbiome and its importance on our overall health.
The microbiome plays an important role in maintaining the skin’s barrier function and plays a crucial part in the development of our immune system, preventing pathogenic bacteria from colonising and infecting the skin. Diabetic patients have been shown to have a distinct microbiome which is less diverse than healthy individuals. This may be due to the changes that occur in their skin such as dryness or increased pH, which undoubtedly affects the microbiome. Having a less diverse microbiome would enable pathogens to take over, activating inflammation and consequently disease.
Infections in chronic DFUs are usually polymicrobial where different bacteria would be living in a complex system forming a biofilm. Commensal bacteria, which live harmoniously on the skin, can become opportunistic pathogens when they find themselves in the nutritious and moist environment of wounds. These may then act synergistically with pathogenic bacteria to cause chronic infections in DFUs. The biofilm will make the ulcer impenetrable to the host’s immune system and systemic antibiotics. Inside this biofilm, microbes are continuously communicating and interacting with each other as well as the host.
In order to identify the pathogens that are causing DFU infections, conventional bacteriological culture techniques are normally used. These techniques tend to favour bacteria that are easily cultivable such as Gram negative rods and Staphylococcus spp. but these organisms may not be the most abundant or the most clinically relevant. Treatment would be tailored according to the culture results and may therefore not target the whole microbiome and pathogens that are actually present, leading to treatment failure, antibiotic resistance and further ulcer formation and amputations.
In Malta, treatment and patient management is tailored according to British guidelines. However the climate and lifestyles of these two countries are totally different eluding to the fact that the skin and ulcer microbiome of the two populations may also be different. It is therefore extremely important to study and document the microbiome of the Maltese population which would ultimately reflect changes in treatment strategies.
Advanced sequencing techniques, such as 16S Next Generation Sequencing (NGS) and Whole Genome Sequencing (WGS), has allowed researchers to identify and better understand the complex microbial communities that reside on the skin as well as DFUs. This will undoubtedly lead to better targeted and personalised antibiotic therapy based on ones’ genome and pharmacogenomic profile.
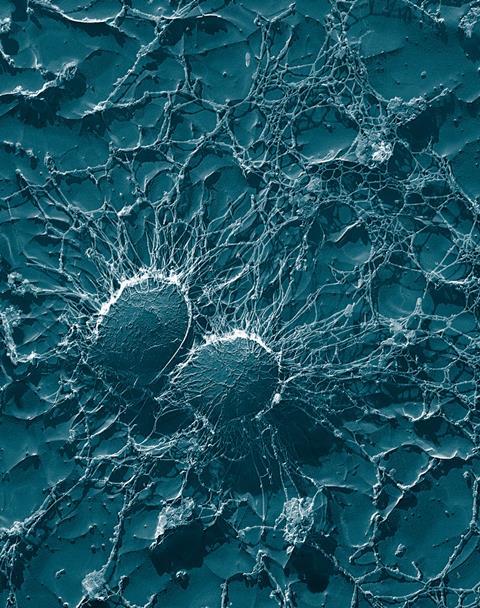
Astronauts and the skin microbiome
The harsh environment of space has many stressors that affect bacteria. Microgravity, temperature changes and high cosmic radiation are examples of such stressors which would trigger genetic changes in bacteria such as those leading to antibiotic resistance and increased virulence, in order for them to adapt and continue to thrive. Understanding the mechanisms of these changes and how bacteria behave in space is crucial for the astronaut’s wellbeing and overall health as one wouldn’t wish to develop infections with multi-drug resistant organisms when in deep space away from medical assistance and intervention.
Space also takes its toll on humans. The astronaut’s skin, as in diabetic patients, gets thinner and drier over time which would make it itchy. Scratching, as well as the constant rubbing of the spacesuit when outside the ISS, would cause damage to the skin facilitating wound formation. Water supplies onboard the ISS are limited and therefore good hygiene is also difficult to maintain. Over prolonged space missions, astronauts have also been shown to have their immune and circulatory systems perturbed making them prone to infections and preventing immune cells from reaching the wound. Astronauts that spend more than a year in space experience a shift in their skin microbiome with a reduction in Gram negative bacteria and an increase in Firmicutes, which may continue to exacerbate skin hypersensitivity and infections by opportunistic pathogens.
Project Maleth
Conducting space bioscience experiments on DFU samples and investigating their genetic composition by molecular techniques, not only helps in identifying the microbiome present in DFUs but also allows a better understanding of how this microbiome adapts to the harsh environment of space. Having a better insight of the DFU microbiomes, as well as their mutations in response to the stresses of space, would undeniably lead to better patient treatment and management improving clinical outcomes but are also of utmost importance for future space exploration.

The experiment
Project Maleth, which is composed of three space missions, is part of a larger study on diabetic foot ulcers involving over 100 diabetic patients. For Project Maleth I, six tissue samples from DFUs of six different patients were surgically removed during amputation surgery and sent to our laboratory. Each tissue was cut into several pieces and processed both for culture, using the conventional methods (currently applied in our hospital laboratories) and molecular testing.
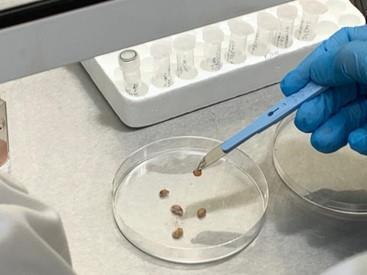
Two of these tissue pieces were placed in cuvettes with phosphate buffered saline, one piece of tissue was destined to the ISS whilst the other piece of tissue would serve as the Earth analogue experiment.

The six pieces of tissues were then shipped to Space Application Services in Belgium where they were loaded onto the International Commercial Experiment (ICE) Cube which in turn was then shipped to the Kennedy Space Centre and readied for launch.

The ICE Cube was then loaded onto the SpaceX CRS23 Cargo Resupply mission contracted by NASA and was lifted off on 29 August, 2021.


Whilst on board the ISS we had the facility to monitor some telemetry such as the temperature. This was important as it enabled us to keep the earth control samples at the same temperature reducing variables that may affect the experiment. We could also monitor the experiment visually with the help of two cameras incorporated in the ICE Cube itself.

After 30 days in orbit, the experiment was unloaded from the ISS by the astronauts on board and sent back to Earth on the same Dragon vessel. Once the samples were returned back to our labs we then processed both the ISS and Earth control samples by cultures and 16S NGS.
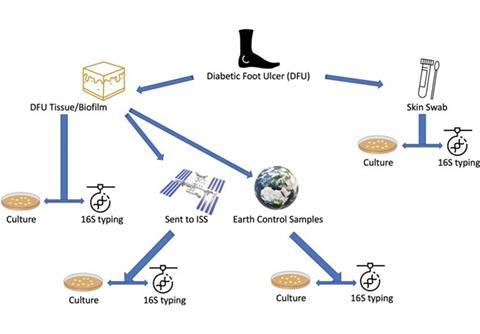
Experiment workflow for Project Maleth, taken from https://pubmed.ncbi.nlm.nih.gov/36544819/
This experiment was again repeated the following year for Project Maleth II, where five tissue samples from DFUs were again sent to the ISS for a month on the 15 July, 2022 on the SpaceX CRS 25 Cargo Resupply mission.

From the preliminary 16S NGS results performed on Project Maleth I samples it was evident that there was a higher relative abundance of organisms in ISS samples compared to the Earth control samples. The most common Gram negative pathogens that were in higher abundance in the ISS samples were Proteus mirabilis and Morganella morganii which are both known DFU pathogens. With regards to culture results, Gram positive organisms that were cultured from the original tissue samples were not cultured after 30 days for both the ISS and earth control samples. One possible explanation might be that PBS was not the ideal medium. Remarkably however, Pseudomonas aeruginosa, Stenotrophomonas maltophilia and Morganella morganii were cultured from the majority of the ISS samples. It appears that Gram negative rods thrive better in space, however the exact mechanism and reasons as to why remain yet to be seen.
What’s next?
The third and final experiment, Project Maleth III has been launched on 15 March 2023, and is therefore currently in orbit when this feature is published. Three tissue samples from DFUs and three skin scrapings from healthy controls have been incorporated into the ICE Cube and form part of this experiment. These samples will also remain in orbit for 30 days and on their return they will be analysed by culture and 16S NGS as the previous Project Maleth experiments.

Project Maleth’s next objective is to look at the full genomic profile of each microorganism present in the ISS and Earth control samples and determine if there are any important nucleotide differences leading to antibiotic resistance or increased virulence. We shall also perform metabolomic analysis in order to determine any metabolites that were excreted and produced during their time in space.
On a larger scale, WGS and metabolomic analysis will also be performed on all the skin and tissue samples obtained from over 100 diabetic patients suffering from DFUs to determine the microbiome composition of Maltese patients.
We aspire that by analysing the DFU microbiome in detail and how this behaves in space, new insights into the prevention and treatment of this debilitating complication of diabetes will be obtained, ultimately improving the quality of life for those affected and for future space inhabitants.
Stephen Hawking once said ‘remember to look up at the stars and not down at your feet’…well we are aiming to do both! With feet firmly grounded to allow us to do the research we all love best, whilst simultaneously looking at the stars for inspiration and planning our next set of experiments to be sent to low earth orbit, or who knows? Even deeper into space!

You can read the author’s paper on The Maleth program here








No comments yet