Strawberry fruit color is primarily determined by anthocyanins, pigments synthesized through a tightly regulated metabolic pathway. Central to this process is the transcription factor MYB10, which activates multiple genes required for pigment production.
Unlike diploid crops, the cultivated strawberry carries eight sets of chromosomes, meaning most genes exist as multiple homoeologous copies with unequal contributions to gene expression.
This genomic redundancy has long complicated both functional studies and breeding efforts, as disrupting the wrong copy may yield no visible effect. Natural white-fruited strawberries suggest that specific MYB10 variants are decisive, but directly proving this in elite cultivars has remained challenging. Based on these challenges, there is a strong need to precisely dissect and manipulate dominant gene copies in polyploid crops.
Researchers from Kongju National University, the National Institute of Horticultural and Herbal Science of Korea, Chungbuk National University, and the University of Florida report a new development in precision fruit trait engineering.
In a study published (DOI: 10.1093/hr/uhaf272) in Horticulture Research in 2025, the team used CRISPR/Cas9 to selectively edit a single dominant gene copy controlling fruit color in the commercial octoploid strawberry cultivar ‘Florida Brilliance’. By targeting the MYB10-1B gene, they successfully converted red strawberries into stable white-fruited plants, demonstrating unprecedented control over a complex polyploid genome.
Anthocyanin regulation
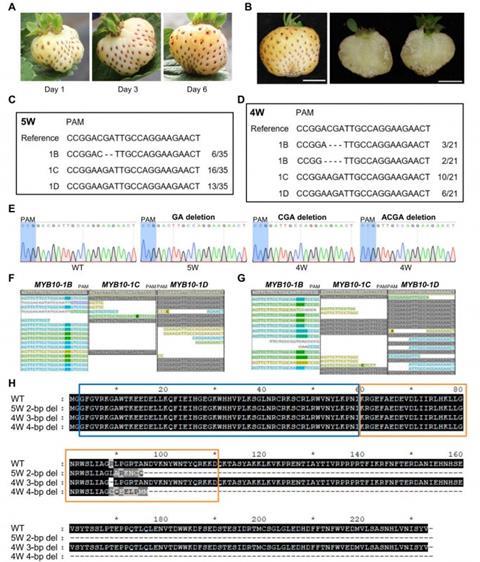
The researchers first confirmed that among several MYB10 gene copies in octoploid strawberry, MYB10-1B is the transcriptionally dominant regulator of anthocyanin accumulation during fruit ripening. Transcriptome analyses showed that this specific homoeolog is highly expressed in red fruits, while other copies contribute minimally and do not compensate when MYB10-1B is disrupted. Guided by this insight, the team designed a CRISPR/Cas9 strategy that exploits subtle sequence differences between homoeologs, allowing precise editing of MYB10-1B while sparing closely related copies.
Edited plants were rigorously screened using high-resolution melting analysis, targeted amplicon sequencing, and whole-genome resequencing. The resulting white-fruited lines carried either homozygous or biallelic mutations exclusively in MYB10-1B, with minimal off-target effects detected elsewhere in the genome. Importantly, the edited fruits remained white throughout development, while the seeds retained their red pigmentation, revealing tissue-specific regulatory mechanisms.
Strong down-regulation
Gene expression analyses further showed that disruption of MYB10-1B caused strong down-regulation of key anthocyanin biosynthesis genes, including CHS, DFR, and ANS. Together, these results establish MYB10-1B as the decisive genetic switch for strawberry receptacle color and demonstrate that precise homoeolog-specific editing is both feasible and essential for trait manipulation in polyploid crops.
“This work shows that in polyploid crops, precision matters more than power,” said the study’s corresponding author. “Rather than knocking out all gene copies, we demonstrated that editing a single, dominantly expressed homoeolog is enough to drive a dramatic and stable phenotypic change. This approach reduces unintended effects and opens new possibilities for functional genomics and breeding in crops with complex genomes. It provides a practical roadmap for translating genomic knowledge into real-world trait improvement.”
Fine-tuning quality traits
Beyond creating visually striking white strawberries, this study has broad implications for crop improvement. The demonstrated strategy can be applied to fine-tune other quality traits such as flavor, nutritional content, and stress tolerance in polyploid fruits and vegetables.
By enabling targeted modification without disrupting redundant gene copies, homoeolog-specific genome editing could accelerate breeding while preserving elite genetic backgrounds.
More broadly, the work underscores how advances in high-quality reference genomes and precision CRISPR design are transforming polyploid crops from genetic challenges into opportunities for innovation, offering breeders new tools to meet consumer demand and agricultural sustainability goals.
Topics
- Agriculture
- Asia & Oceania
- Bioengineering
- Chungbuk National University
- CRISPR/Cas9
- Food Security
- Healthy Land
- Kongju National University
- Microbial Genetics
- MYB10-1B
- National Institute of Horticultural and Herbal Science of Korea
- polyploid genomes
- precision fruit trait engineering
- Research News
- Soil & Plant Science
- strawberry plants
- University of Florida
- USA & Canada







No comments yet