Researchers have discovered a tiny molecular switch that can control and reverse the direction of a rare rotary machine linked to motility, surface colonization and protein secretion in bacteria.
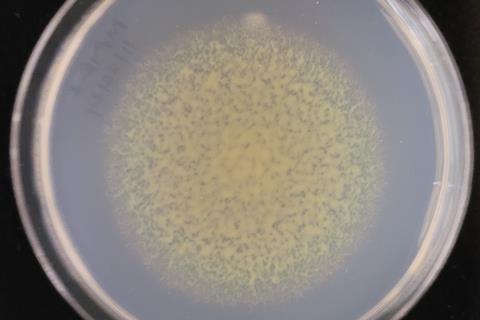
In a study published in mBio in June 2025, Abhishek Trivedi and colleagues at Arizona State University discovered that a protein called GldJ acts like a molecular switch that can change the direction of the Type IX Secretion System (T9SS).
This matters because T9SS helps certain bacteria move across surfaces and export proteins that are important for survival, colonization, and interaction with their environment.
Complex molecular machines
“Many bacteria depend on complex molecular machines to move, colonize surfaces, and release proteins that help them survive. Some of these bacteria are important in the environment, while others are relevant to animal or human health,” said Dr Trivedi.
“The Type IX Secretion System is one of the most unusual of these machines because it is involved in both protein secretion and surface motility, but scientists still do not fully understand how it works.
“In our work, we wanted to understand how this system controls its direction of movement of the tiny rotary motor. In simple terms, we were asking: what makes this tiny bacterial machine rotate one way rather than the other?”

The team studied the gliding bacterium Flavobacterium johnsoniae, one of the best model organisms for understanding bacterial gliding motility and how the Type IX Secretion System (T9SS) works. They focused on a protein called GldJ, which is associated with a moving cell-surface “conveyor belt” that helps the bacterium glide.
Eight amino acids
Dr Trivedi made a precise genetic change by deleting a short region at the end of the GldJ protein. He then used bacterial genetics, motility assays, microscopy, and molecular simulations to test how that change affected the system. What he found was striking: removing just eight amino acids from GldJ caused the rotational direction of the system to switch from mostly counterclockwise to clockwise.
In other words, GldJ was not just part of the machinery; it turned out to be a control point for the direction of the motor. The study also identified changes in another protein, GldK, suggesting that the interaction between GldJ and GldK is an important part of how this direction-switching works.
“The most surprising result was that deleting only a short region of GldJ was enough to reverse the direction of the motor. We expected the mutation might disrupt movement but finding that it could actually switch the direction of rotation was unexpected and revealed a new level of control in the system,” Dr Trivedi said.
“This work helps explain how bacteria control a specialized machine used for both movement and protein export. That matters because these systems influence how bacteria colonize surfaces, adapt to changing environments, and interact with hosts.
Next steps
“T9SS-related systems are found in bacteria relevant to the oral microbiome, the gut microbiome, and other important environments. By revealing a molecular switch that controls direction, my study gives researchers a new way to think about how bacteria fine-tune their behavior and may eventually help guide work on how to interfere with harmful bacterial processes.
“The next step is to understand in more structural detail how GldJ interacts with GldK and the rest of the machinery during movement. We also want to know how widespread this control mechanism is in other bacteria that use the Type IX Secretion System. Over time, that could improve our understanding of how similar bacterial systems contribute to colonization, persistence, and secretion in different environments.”
READ MORE: Secrets of microbial motion: How bacteria swash, glide and shift gears to survive
READ MORE: Tiny hinges bend the infection-spreading spikes of a coronavirus
The study was carried out by a team at Arizona State University. Abhishek Trivedi, the paper’s first author, led the experimental work, including generating the mutants and performing the cell-tethering and TIRF microscopy assays. Collaborators contributed motility analysis and molecular dynamics simulations. The paper states that Abhishek Shrivastava and Abhishek Singharoy supervised key parts of the work, and the study was supported by NIH and NSF funding.






No comments yet