Antivirals given for chronic hepatitis B include nucleos(t)ide analog monotherapy and pegylated interferon-alpha therapy, but the safety of interferon in patients with cirrhosis has been unclear.
A study in Portal Hypertension and Cirrhosis analyzed data from two large Chinese cohorts, including 920 patients with compensated cirrhosis, and found that interferon therapy was not associated with a higher risk of serious complications, supporting its use with appropriate monitoring.
Hepatitis B is a viral infection that affects the liver and can lead to a chronic diseased stage. Antiviral treatments aim to reduce complications such as liver failure and liver cancer, making long-term disease control a key goal. While hepatitis B can be prevented with a vaccine administered soon after birth, individuals with chronic infection are typically treated with antiviral medications.
Currently, the main treatments for chronic hepatitis B include nucleos(t)ide analog (Nuc) monotherapy and pegylated interferon-alpha (Peg-IFN-α) therapy. IFN-based treatments are widely used, especially in Asian populations, and some studies have linked them to a comparatively long-lasting control of the virus and a lower risk of liver cancer.
However, the use of IFN-based therapy in patients with cirrhosis remains debated due to concerns that it may be associated with worsening liver function or increased side effects, such as low platelet counts and liver abnormalities.
Safety of therapy
To examine this further, a team of researchers in China led by Professor Wenhong Zhang of Huashan Hospital, Fudan University, and Professor Xiaolong Qi of Zhongda Hospital, Southeast University, evaluated the safety of Peg-IFN-α therapy in patients with chronic hepatitis B and compensated cirrhosis. Their findings were published online in the journal Portal Hypertension & Cirrhosis on March 06, 2026.
“We assessed the safety of Peg-IFN-α therapy in patients with compensated hepatitis B cirrhosis by evaluating whether it led to higher decompensation events, more severe adverse events, or worse outcomes compared to nucleos(t)ide analog therapy (the standard treatment for hepatitis B), or to patients without cirrhosis,” the authors note.
The researchers analyzed data from two large studies conducted in China. One, the OASIS Project, followed patients with chronic hepatitis B to understand how different antiviral treatments perform in real-world settings. The other, CHESS 2306, focused on patients with compensated cirrhosis receiving IFN-based therapy between 2018 and 2024. The study included adults aged 18 to 65 who had been on treatment for at least 12 weeks.
In total, the analysis included 920 patients with Child-Pugh class A hepatitis B cirrhosis who received IFN therapy. This stage refers to an early compensated stage of cirrhosis in which the liver is still functioning relatively well. These patients were compared with those receiving Nuc therapy and those without cirrhosis, with outcomes assessed over up to 48 weeks.
Studying side effects
The researchers examined serious complications such as liver decompensation, which includes fluid buildup, bleeding, and brain-related symptoms, as well as other side effects. These complications were rare and occurred at similar rates in both IFN and standard treatment groups, with no treatment-related hospitalizations or deaths reported.
Severe side effects, such as low blood cell counts and liver-related abnormalities, were also similar between groups. When comparing groups with and without cirrhosis who received IFN, most side effects were comparable. However, patients with cirrhosis who already had elevated bilirubin levels were more likely to experience further increments during treatment.
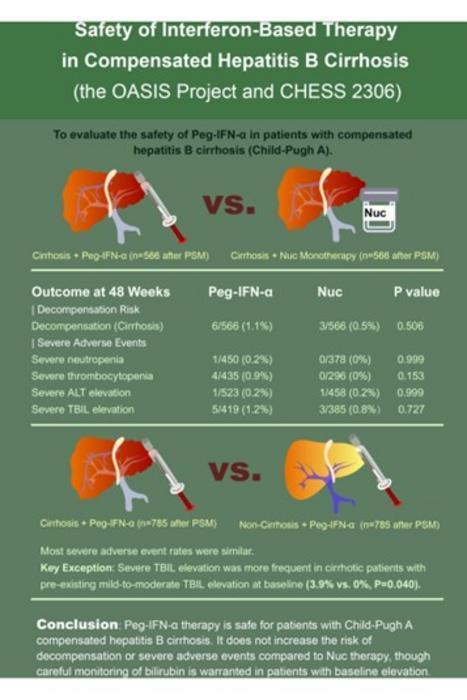
Overall, the results suggest that IFN-based therapy was generally safe for patients with compensated cirrhosis. The researchers did not find it to be associated with a higher risk of serious complications compared to standard treatment; however, milder side effects were more common and required closer monitoring. The authors note that careful patient selection and follow-up remain important, and that IFN-based therapy should be used with caution in patients with cirrhosis and elevated bilirubin levels.
Patient selection
“The findings of this study indicate that IFN‐based therapy has acceptable safety in a population with Child-Pugh class A cirrhosis. However, in cirrhotic patients with CHB and abnormal TBIL levels, IFN administration is more likely to lead to severe TBIL level elevation,” the study reports.
IFN-based therapy is a promising strategy for achieving long-term viral control and reducing liver cancer risk in chronic hepatitis B. These findings offer encouraging evidence on the safety of IFN-based therapy in patients with compensated cirrhosis and may help inform treatment decisions.




No comments yet