Cryptococcus neoformans is one of four fungi classified as ‘critical priority’ on the WHO’s Fungal Pathogens Priority List, which was published in October 2022 following decades of research and calls for fungal pathogens to be classified alongside their bacterial and viral counterparts.
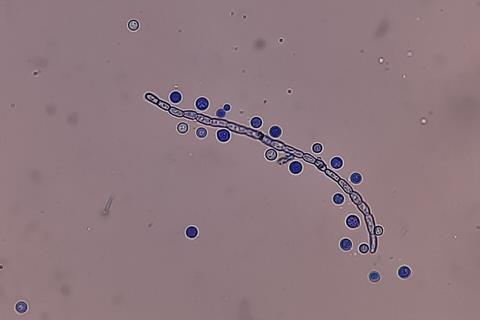
The fungus infects people through inhalation of spores or yeast cells in the environment, first colonising the lungs and can then spread to the brain. In 2020, an estimated 112,000 deaths were associated globally with fungal meningitis caused by C. neoformans.
Increasing evidence shows that co-infection of Mycobacterium tuberculosis, the causative agent of tuberculosis, together with C. neoformans, is a grave public health concern, increasing the risk of death significantly compared to fungal infection alone.
“We know that these pathogens are commonly recovered from patients in endemic regions, but no one has investigated the impact of one on the other,” says Orlando Ross, first author of the study and PhD candidate at the MRC Centre for Medical Mycology at the University of Exeter, UK. “We hypothesised that co-incubation of C. neoformans with Mycobacterium species would result in C. neoformans cells changing in size and shape, towards a more pathogenic and dangerous form.”
Changes in presence of Mycobacteria
What they found confirmed their thinking: in the presence of Mycobacteria, the fungus changed its cell density, cell diversity, and capsule size – a protective outer membrane that surrounds the cells of the fungus. Some of these changes are known to increase the fungus’ harmfulness to its infected host, pointing to how co-infection could lead to worse clinical outcomes.
READ MORE: Discovery could lead to new treatments for drug-resistant fungal infections
READ MORE: Cannabis extracts show remarkable effectiveness against fungal pathogens
To further their understanding, the researchers also tested how this might impact the immune system during co-infection. By replicating the co-infected lung environment, they uncovered that immune cells were more likely to be invaded by C. neoformans when they were exposed to tuberculosis, compared to when they were not.
“This shows that the presence of both bacterial and fungal pathogens together in the lung could worsen disease outcome for patients,” Ross explains.
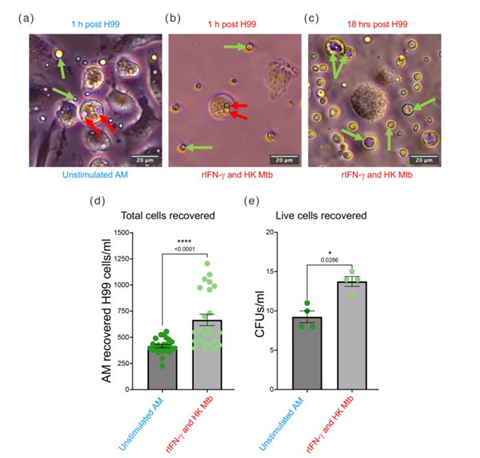
The findings raise further questions about how this can be used to improve clinical outcomes in the future for those doubly infected. The next step will be modelling these co-infections in mouse models. “We’re really proud of the physiological modelling we’ve carried out in this project, but we’re excited to validate these findings in the context of a full immune system and more complex host environments,” says Ross.







No comments yet