A new kind of CRISPR that destroys cells rather than gene editing them has shown potential for killing sick cells while leaving healthy cells untouched.
CRISPR-mediated gene editing technology has revolutionized science and medicine, showing promise for fixing the root cause of genetic diseases. One of the most well-known CRISPR proteins, Cas9, acts like a pair of molecular scissors to make precise DNA edits.
Now, a new technology is deploying a recently discovered CRISPR protein, Cas12a2, which acts like a paper shredder. When activated by its target, it rips the genome apart, a lethal move that researchers can program to destroy harmful virus-infected cells or cancer cells.
“For as long as medicine has been practiced, a significant challenge has been: how do we eliminate harmful cells without damaging healthy ones?” says Jared Thompson, graduate researcher in biochemistry at University of Utah Health and one of the co-first authors of the paper. “This is the focus of so much biomedical research in the world today, and Cas12a2 is a very promising tool for exploring that.”
The results are published in the journal Nature.
“Its goal is to destroy anything it sees”
In the most common kind of CRISPR gene editing, a protein called Cas9 recognizes a specific DNA sequence and makes a single precise cut there. Like regular CRISPR, Cas12a2 recognizes a very specific genetic sequence—within an intermediate gene product called RNA rather than DNA itself. Like regular CRISPR, researchers can easily program which specific sequence activates Cas12a2. But once Cas12a2 recognizes its target, it starts cutting DNA and doesn’t stop.
“Its goal is not to correct anything,” says Yang Liu, PhD, assistant professor in biochemistry at U of U Health and one of the co-senior authors of the paper. “Instead, it’s to destroy anything it sees.”
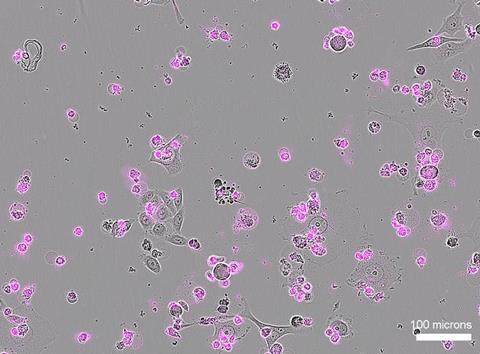
Overwhelmed by DNA damage, targeted cells self-destruct. This might sound more dangerous than therapeutic. But researchers can easily target the molecule to be activated by RNA sequences that are specific to cancer cells or dangerous viruses.
Cancer-killing CRISPR
The researchers targeted Cas12a2 to a cancer mutation in a gene called KRAS. The mutation can cause cancerous overgrowth. Cas12a2 reduced the growth of human lung cancer cells with this mutation in a dish by 50%—working about as well as established anticancer drugs like cisplatin.
But unlike chemotherapy drugs, Cas12a2 didn’t affect cells with healthy KRAS at all.
“The enzyme that we’re working with is extremely specific,” Liu says. “It does not touch the healthy cells. So if we’re thinking about a cancer therapy, you’re treating cancer with no side effects. That was striking to us. We did not know that was possible.”
Targeting infectious disease
Cas12a2 was also able to efficiently kill cells infected by human papillomavirus (HPV, which can cause genital warts or cervical cancer) when the researchers targeted it to a viral RNA. Collaborators at Akribion Therapeutics found that Cas12a2 reduced the growth of infected cells in a dish by more than 90% without harming healthy cells.
Injecting HPV-targeted Cas12a2 into virus-infected tumors in mice also slowed tumor growth, showing that the strategy can work in animal models of disease. Liu says that Cas12a2 could also be easily programmed to target other viral diseases, including HIV.
The path forward
There’s a long way to go before Cas12a2 could be used in human therapies. The large majority of the Cas12a2 tests so far have been done in cells in a dish; more research is needed to test efficacy and safety with animal models before clinical trials with patients become a possibility.
“If you try to treat an organism, different organ systems might uptake Cas12a2,” Thompson cautions, “and we don’t yet know how just the presence of the protein, even if it’s not being activated, affects an organism.” The researchers add that, in addition to safety concerns, delivering enough Cas12a2 to the parts of the body that need it will be a fundamental challenge.
Still, the researchers are optimistic. Besides treating viral infections and cancer, Liu also thinks the molecule’s cell-killing potential could be applied to treat neurodegenerative diseases by eradicating cells in the brain that produce toxins. Or the molecule could be used to reverse other aging-related diseases by killing sick cells that consume resources without contributing function.
What’s next for this research? Liu says: “Curing the incurables.”
Background
This work was made possible by collaboration between researchers at University of Utah Health, Huntsman Cancer Institute at the University of Utah, Akribion Therapeutics, BRAIN Biotech AG, the Helmholtz Institute for RNA-based Infection Research, and the University of Würzburg. In addition to Thompson and Liu, Paul Scholz (co-first author), Kadin Crosby (co-first author), Torsten Fauth, Nathan Krah, Grant Schlauderaff, Robin Back, Zachary Berkheimer, Alivia Jolley, Dirk Sombroek, Rebekka Medert, Christian Zurek, Oleg Dmytrenko, Emily Wilson, Friso Schut, Jared Rutter, Xiaoyang Zhang, Michael Krohn, Ryan Jackson (co-senior author), and Chase Beisel (co-senior author) are authors on this paper.
This research was supported by the R. Gaurth Hansen Family, the European Research Council, the HSC Cell Imaging and Flow Cytometry Cores at the University of Utah, the High-Throughput Genomics and Cancer Bioinformatics Shared Resource at Huntsman Cancer Institute at the University of Utah, and the National Institutes of Health, including the National Cancer Institute (P30CA042014) and the National Institute of General Medical Sciences (R35GM138080, R35GM150941, and T32GM141848). Content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Scholz, Zurek, Krohn, Dmytrenko, Jackson and Beisel have pending patent applications and awarded patents related to the presented work. Beisel is a co-founder and scientific advisor to Locus Biosciences, a co-founder and officer of Leopard Biosciences GmbH, and scientific advisor to Benson Hill. Zurek is an employee of BRAIN Biotech AG; Scholz, Fauth, Back, Sombroek, and Medert are employees of Akribion Therapeutics GmbH. Krohn is a co-founder and co-CEO of Akribion Therapeutics GmbH. The other authors declare no relevant conflicts of interest.







No comments yet