Patients with severe acute pancreatitis often experience disrupted intestinal barrier function, extensive antibiotic exposure, and prolonged ICU treatment, all of which increase vulnerability to secondary infections. These infections—particularly respiratory and bloodstream infections—are a major contributor to mortality and healthcare burden.
While probiotics have been proposed as a way to restore microbial balance, prior clinical trials in critically ill patients have yielded conflicting results, and concerns about safety have limited their adoption. Most existing studies focus primarily on bacteria, leaving the role of intestinal fungi largely unexplored. Based on these challenges, there is a clear need to investigate whether targeted modulation of both bacterial and fungal microbiota can safely reduce nosocomial infections in severe pancreatitis.
Researchers from Xinqiao Hospital, Army Medical University (China) report new clinical evidence that a fungal probiotic may reduce hospital-acquired infections in patients with severe acute pancreatitis. The findings were published (DOI: 10.1093/burnst/tkag006) in 2026 in Burns & Trauma. In a randomized controlled trial conducted in an intensive care setting, patients receiving Saccharomyces boulardii alongside standard enteral nutrition experienced significantly fewer infections than those receiving nutrition alone. The study also tracked dynamic changes in respiratory and intestinal microbiota, offering mechanistic insight into how probiotic intervention may influence infection risk.
Fungal probiotic
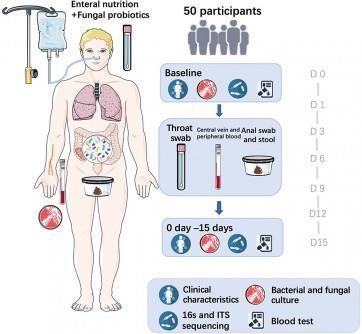
The single-center, open-label randomized trial enrolled 50 patients with severe acute pancreatitis admitted to a gastroenterology ICU. Participants were assigned to receive either standard enteral nutrition alone or nutrition supplemented with the fungal probiotic Saccharomyces boulardii for 15 days. Researchers longitudinally collected throat and rectal swabs to monitor bacterial and fungal communities using full-length 16S rRNA and ITS sequencing.
Clinically, none of the patients receiving the probiotic developed nosocomial infections, compared with more than one-fifth of patients in the control group. Respiratory infections and catheter-related bloodstream infections occurred exclusively in the control group. Importantly, probiotic use did not increase the risk of fungemia, addressing a key safety concern.
Probiotic supplementation
Microbiome analyses revealed that probiotic supplementation reduced intestinal dysbiosis, suppressed overgrowth of Enterococcus in the gut, and limited Candida proliferation in both the respiratory tract and intestines. Statistical modeling further identified intestinal fungal burden as a strong predictor of infection risk, with fungal indices showing high accuracy in predicting nosocomial infections. Together, these results suggest that fungal probiotics may help stabilize the gut–lung microbial axis, thereby reducing infection susceptibility in critically ill pancreatitis patients.
“The findings highlight the overlooked role of fungal communities in critical illness,” said the study’s senior investigators. “While most infection-prevention strategies focus on bacteria, our results show that intestinal fungi are closely linked to infection risk in severe pancreatitis. By selectively suppressing harmful microbial overgrowth without disrupting overall diversity, fungal probiotics may offer a safer, more targeted approach than broad antimicrobial strategies. These insights open the door to microbiome-informed interventions in intensive care medicine.”
Low cost strategy
If confirmed in larger, multicenter trials, fungal probiotic supplementation could become a low-cost, adjunctive strategy to reduce hospital-acquired infections in severe pancreatitis. Beyond pancreatitis, the findings may have broader implications for other critically ill populations where microbiome disruption drives infection risk.
READ MORE: Studying yeast in the gut could lead to new, better drugs
READ MORE: Probiotic delivers anticancer drug to the gut
The identification of fungal biomarkers as predictors of nosocomial infection also points toward new tools for early risk stratification. More broadly, the study supports a shift toward precision microbiome management in intensive care—balancing microbial suppression with ecosystem preservation to improve patient outcomes.







No comments yet