Serious fungal infections are on the rise, and many hospital-acquired cases are becoming harder to treat as fungi become resistant to available medications.

One of the most widely used therapies, caspofungin, combats Candida infections that can turn deadly in people with weakened immune systems. Despite the drug’s importance, scientists have never fully understood how it works at the molecular level — or why it sometimes stops working.
A new study in Nature from Duke biochemists Seok-Yong Lee, PhD, and Kenichi Yokoyama, PhD, has now filled in those gaps, offering the most detailed picture yet of caspofungin in action. The findings reveal a surprising mechanism and provide critical insights that could help guide the design of next-generation antifungal drugs.
“By revealing how a frontline antifungal drug actually works and how resistance develops, this study provides a new framework for designing better therapies to combat the growing threat of fungal disease,” Yokoyama said.
An unexpected interaction
Scientists have long believed that caspofungin binds directly to a key fungal enzyme responsible for building the cell wall — essentially shutting down the assembly line fungi need to survive. But the Duke team discovered the real story is more complex.
Caspofungin does not simply attach to the enzyme. Instead, it binds to the growing chain of cell-wall material the enzyme is producing. The drug forms a three-way complex with the enzyme and its polysaccharide product. This interaction jams the enzyme mid process, preventing it from adding new building blocks and effectively stalling cell wall construction.
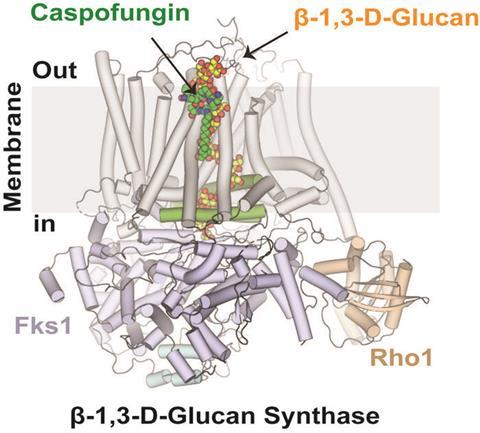
This discovery helps explain how certain mutations enable fungi to escape the drug’s effects — and why caspofungin sometimes fails in the clinic.
Seeing the enzyme in unprecedented detail
Capturing this mechanism required an unusually close look at fungal cell-wall machinery. This challenge required collaboration between Yokoyama’s and Lee’s laboratories. By combining their strengths in enzymology and structural biology, the team tackled a long‑standing problem in antifungal research: how to capture a crucial enzyme while it is actively doing its job.
The researchers developed a method for preparing the enzyme β-1,3-D-glucan synthase in its active state. They paired this imaging with parallel laboratory tests to make sure what the structural snapshots matched how the enzyme behaves in real life.
Critical hurdle
This was a critical hurdle; by synchronizing the enzyme’s working conditions with advanced cryo-electron microscopy, the team was able to see exactly what the enzyme looks like when an antifungal drug shuts it down. This led to a key insight: when the enzyme is inactive, the drug does not bind. Only when it’s actively building the fungal cell wall does it allow interaction with the antifungal drug.
“The images from this study show how the enzyme interacts with caspofungin with the growing glucan chain it produces,” Lee said. “This structural detail was essential to uncovering the drug’s true mode of action.”
With invasive fungal infections increasing worldwide and few new antifungal drugs in development, the discovery comes at a critical time.




No comments yet